Small molecule Drugs
Leonurine Sulfate (SCM-198):
Derived from the natural product motherwort, representing a globally novel chemical entity with a new target and new mechanism of action!
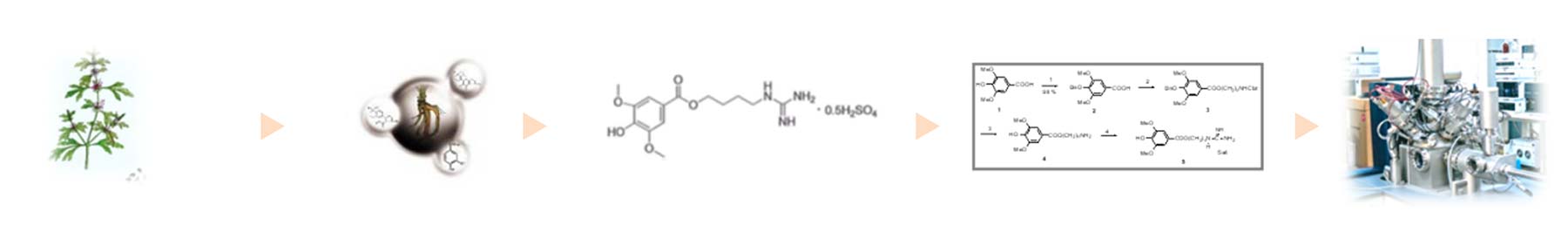
Global First-in-Class:
A first-in-class anti-cardiovascular inflammation drug targeting CD36, filling a global void in this therapeutic target area!
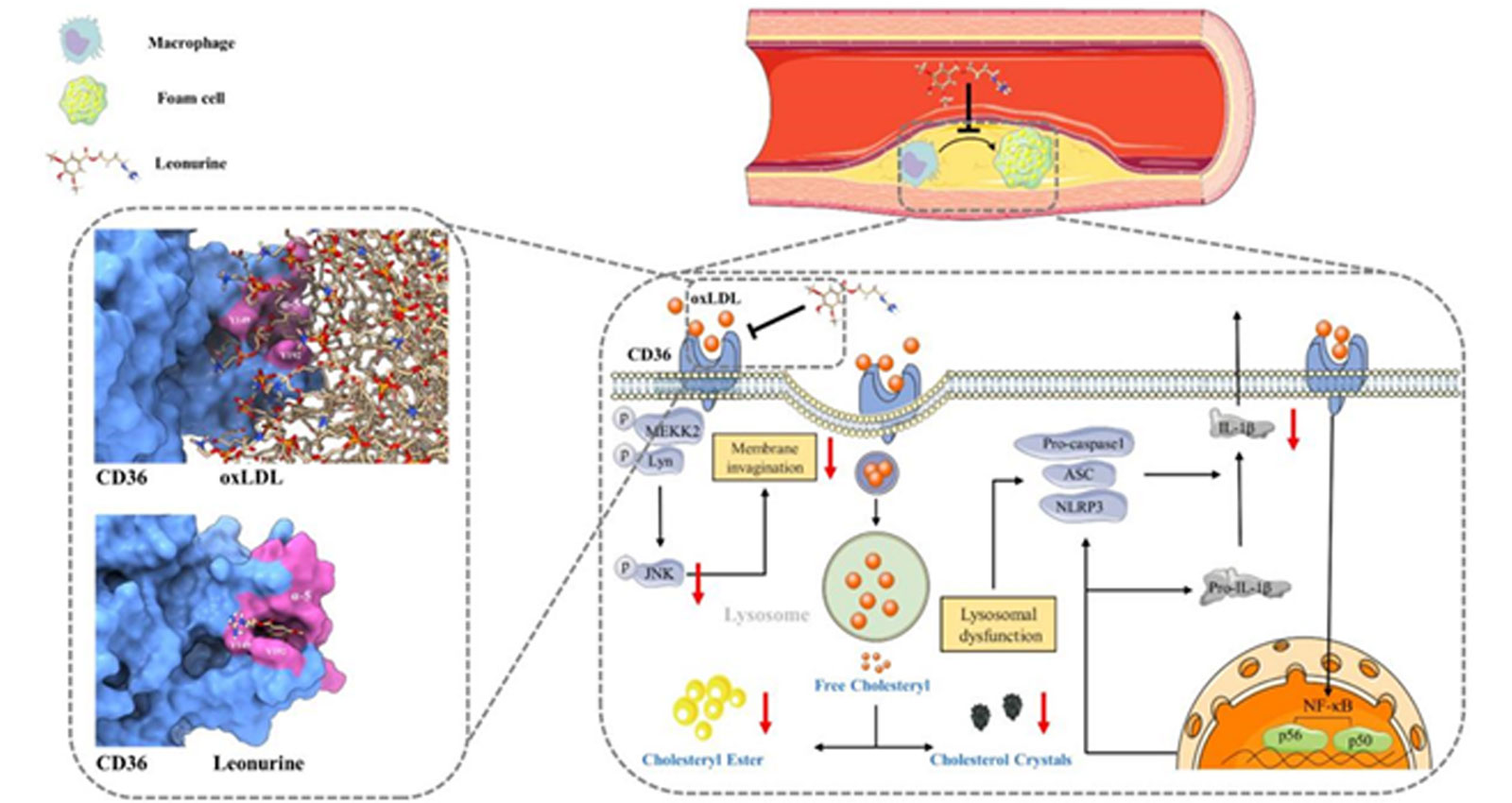
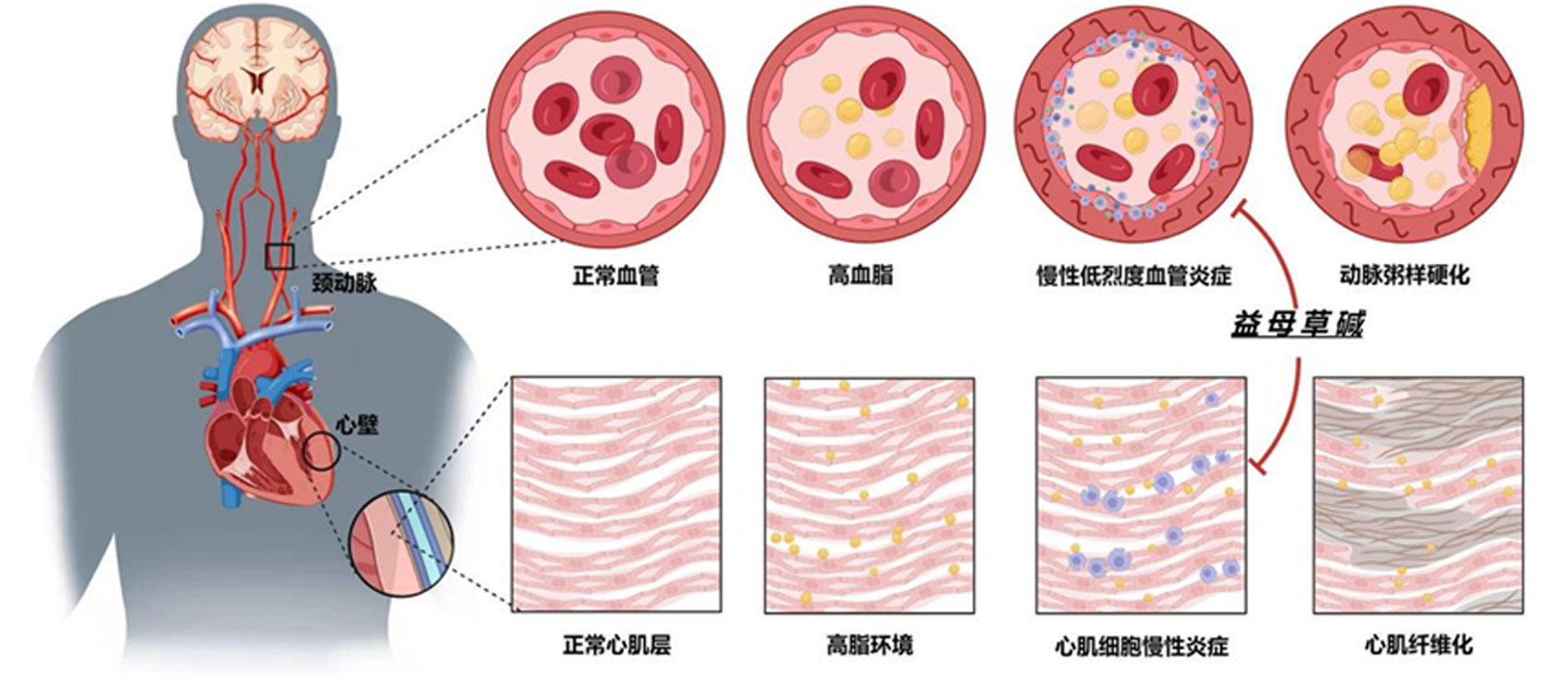
Clinical studies demonstrate that leonurine sulfate (SCM-198) exhibits significant efficacy in treating chronic low-grade vascular inflammation
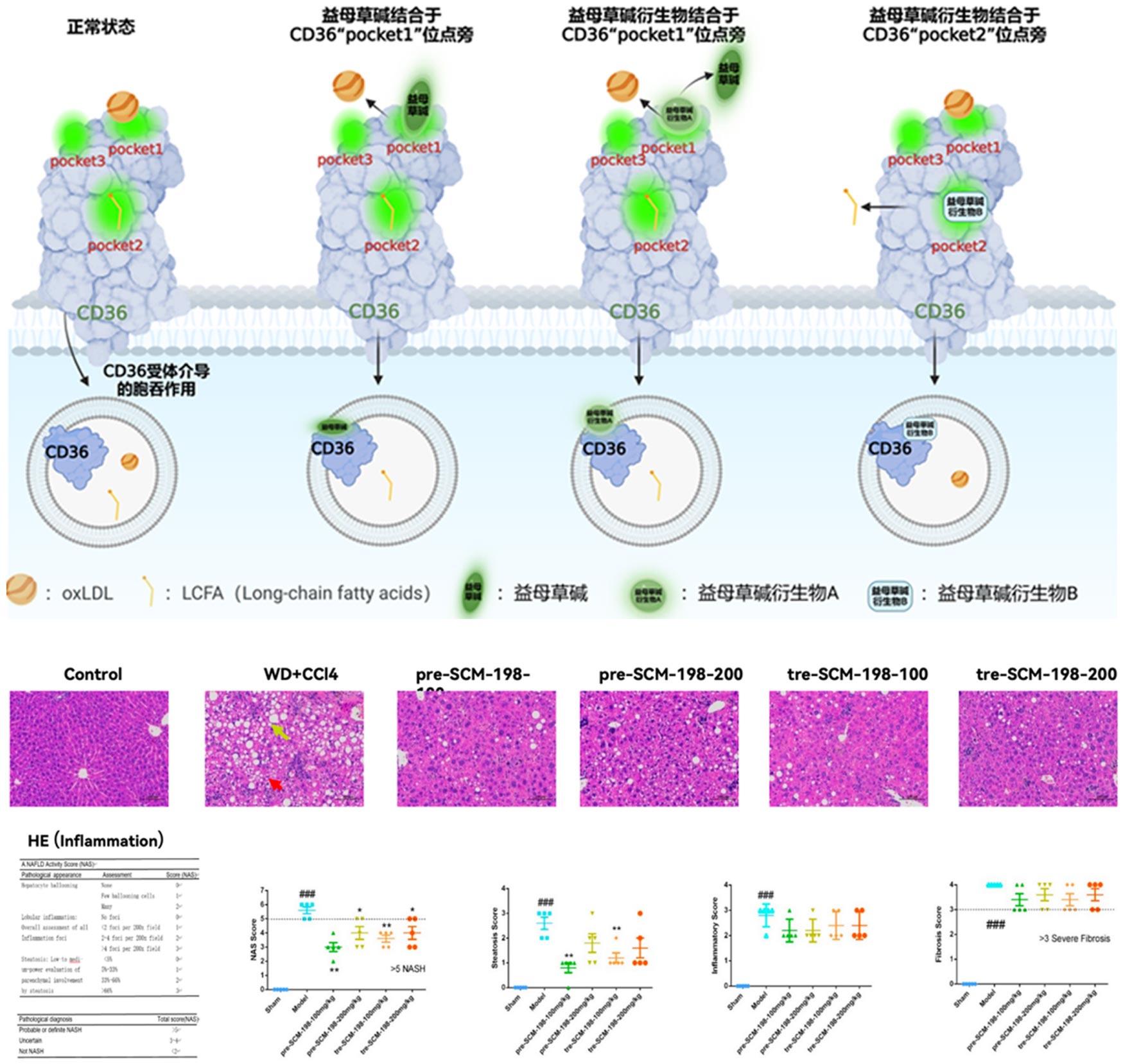
Leonurine sulfate (SCM-198) is the first compound originating from China shown to be effective for non-alcoholic fatty liver disease
Selected Indications Under Investigation
 Hyperlipidemia
Hyperlipidemia
 Non-Alcoholic
Non-AlcoholicFatty Liver Disease
 Atherosclerosis
Atherosclerosis
 Endometriosis
Endometriosis
 Ulcerative
UlcerativeColitis
 Wound Healing
Wound Healing
Leonurine Sulfate (SCM-198) Has Been
Sumitted For Dual China-US Regulatory Filings

National Medical Products Administration of China
Center for Drug Evaluation


U.S. Food and Drug Administration
Intellectual Property
with 1 application pending
Substantial Commercial Value
150 Million RMB
Leonurine sulfate (SCM-198) achieved a successful technology transfer valued at RMB 150 million in 2016
——representing one of the few successful new drug commercialization cases in China

>$200 Million USD
According to authoritative assessments, leonurine sulfate (SCM-198) is projected to achieve an overseas licensing valuation exceeding USD 200 million upon entering FDA Phase II clinical trials!

