Invitation to Tender: SPRC Project
1. Tender Project
This tender is for the “InnoDrug Group Limited SPRC Preclinical Animal Study Project,” focused on non-clinical animal studies for SPRC.
2. Project Overview and Scope of Tender
This project consists of preclinical animal studies for SPRC conducted by InnoDrug Group Limited. The project has completed PCC confirmation. To meet the requirements for a New Drug Investigational New Drug (IND) application in China, the project plans to engage a preclinical Contract Research Organization (CRO) to complete drug safety evaluation research services.
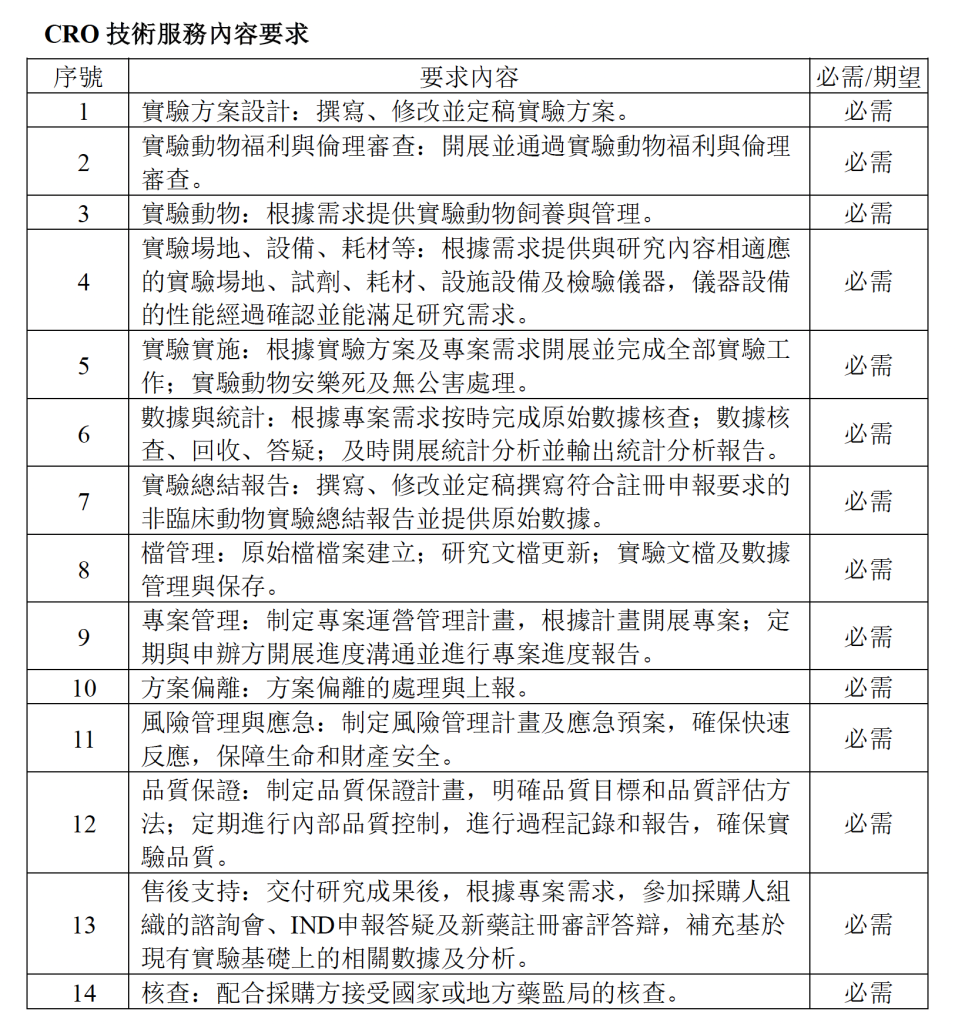
To satisfy the requirements for independent drug registration and marketing approval, this project plans to assess the safety of SPRC through rodent and non-rodent models to determine the safety of clinically relevant parameters. The contracted party shall be responsible for the entire animal study process, including experimental protocol design, organization, implementation, execution, inspection, documentation, archiving, and reporting, ensuring that studies comply with Good Laboratory Practice for Non-Clinical Studies of Drugs (NMPA-GLP), Drug Registration Management Regulations, International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) relevant guidelines, as well as applicable regulations and guidelines issued by the National Medical Products Administration, to ensure that non-clinical animal study results are authentic, complete, accurate, and scientific.
3. Bidder Qualification and Requirements
- 3.1 The CRO company shall possess the necessary qualifications to conduct non-clinical animal studies and demonstrate professional quality assurance capabilities. According to the special requirements for drug registration non-clinical animal studies, the CRO must complete the entire animal study process for this project, including experimental protocol design, organization, implementation, execution, inspection, documentation, archiving, and reporting services. The CRO must ensure that animal studies strictly comply with NMPA-GLP, Drug Registration Management Regulations, and other relevant regulatory requirements, producing research reports and data that meet NMPA registration submission standards to achieve the objective of independent drug registration application.
- 3.2 The CRO company shall thoroughly understand the project requirements and propose specific operational management plans for this project, including but not limited to work implementation plans, quality assurance plans, confidentiality measures, risk management plans, and contingency plans.
- 3.3 The CRO company shall possess hardware and software facilities and equipment that meet project requirements and assemble a professional technical team with extensive experience to implement project services, fully ensuring project quality and efficiency.
- 3.4 The CRO company shall fully cooperate with the sponsor’s experimental requirements. If the sponsor needs to adjust animal numbers or supplement additional experimental projects during project implementation, the CRO company shall actively cooperate in signing supplementary agreements and executing and implementing supplementary studies.
- 3.5 The CRO company shall be able to assign a project director with more than 5 years of toxicology experience to comprehensively oversee the protocol design and project implementation; have research facilities in the client’s location (Shenzhen) with GLP certification; possess AAALAC accreditation; and have access to a toxicologic pathologist with more than 10 years of experience holding Chinese and/or European qualifications.
4. Tender Document Requirements
Specific requirements include:
4.1 Framework of study design from the bidding organization (excluding reproductive toxicity studies).
4.2 Individual study quotations from the bidding organization.
4.3 Overall timeline for the SPRC project.
5. Commercial Requirements
5.1 Commissioned Matters and Cooperation Model
The purchaser commissions the bidder to undertake the non-clinical animal study CRO services project for SPRC. The bidder must, according to the special requirements for drug registration non-clinical animal studies, complete the entire animal study process for this project, including experimental protocol design, organization, implementation, execution, inspection, documentation, archiving, and reporting services, ensuring that studies comply with NMPA-GLP, Drug Registration Management Regulations, and other relevant regulatory requirements, obtaining complete research data to achieve the objective of drug registration application.
5.2 Contract Performance Timeline, Location, and Method
From the effective date of the contract, the project implementation, acceptance, and delivery to the purchaser shall be completed within 8 months after contract signing.
5.3 Acceptance Standards and Methods
5.3.1 Acceptance Standards
In accordance with current national and industry standards, specifications, and technical service requirements for this project.
5.3.2 Acceptance Methods
Upon project completion, the bidder shall deliver technical results to the purchaser, including provision of GLP-compliant comprehensive study reports for this project along with corresponding electronic documents, raw data, other experimental materials and corresponding electronic documents, and materials to be retained by the sponsor. Acceptance shall be conducted through written confirmation by the purchaser in accordance with the experimental protocol for this project, GLP, and relevant regulatory requirements.
5.4 Quality and Intellectual Property Requirements
5.4.1 The bidder shall meet national and industry-relevant quality standards while satisfying the technical service requirements of this project, strictly implementing GLP-related provisions.
5.4.2 Both parties confirm that during project cooperation, intellectual property rights and derivative rights related to equipment and technology used in this project (including but not limited to copyrights, rights to apply for patents, patent rights, patent application rights, trade secrets, trademarks, integrated circuit layout designs, etc.) shall belong entirely to the purchaser, with the bidder having no rights whatsoever.
5.5 Quotation Requirements
5.5.1 The bid quotation shall be based on the CRO service procurement items required in the tender document procurement requirements, including all costs that may occur during protocol design, process management, experimental implementation, output reports, and all work and service processes. The bid quotation shall include all taxes and fees paid or payable by the bidder.
5.5.2 Bidders must respond to the entire package; incomplete bids will be rejected.
5.5.3 Bidders shall provide detailed quotations for each item according to the itemized quotation table. The detailed quotation table serves as a supporting document for the bid. If the quotation conflicts with the itemized quotation table, the itemized quotation table shall prevail.
5.6 Liability for Breach of Contract
5.6.1 The winning bidder shall strictly provide services according to the time and schedule agreed upon with the purchaser. If the actual progress is delayed by more than 30 days compared to the plan due to reasons attributable to the winning bidder, the purchaser has the right to terminate the procurement contract as appropriate and pursue claims against the supplier through legal proceedings. If the winning bidder causes property loss to the purchaser through intentional or gross negligence, the winning bidder shall pay liquidated damages to the purchaser.
5.6.2 The purchaser has the right to review relevant supporting materials (originals) provided by the winning bidder as required by this project. If the winning bidder is found to have provided false materials, the purchaser has the right to recover fees already paid to the winning bidder and require the winning bidder to compensate liquidated damages equal to 30% of the total contract amount. If the liquidated damages are insufficient to cover the purchaser’s losses, the winning bidder shall continue to compensate. All losses resulting from this shall be borne by the winning bidder.
6. Notes and Considerations
6.1 The CRO company must ensure that research content and reports comply with registration application requirements.
6.2 To comply with relevant bidding regulations, the complete set of bid documents shall be sealed, stamped with a valid official seal, and signed by the bidder (otherwise it will be deemed invalid), with the project name displayed on the cover.
6.3 When submitting bid documents, submission may be by mail or direct delivery to the submission location. Failure to submit bid documents within the specified time will be considered as withdrawal from bidding.
6.4 Subcontracting of commissioned work and tasks without authorization is not permitted.
6.5 Bidders shall bear all costs associated with preparing and participating in the bidding process. Regardless of the bidding outcome, there is no obligation or responsibility to bear these costs.