The global community is awaiting China’s next-generation innovative medicines.
This July, China’s pharmaceutical community was ignited by a blockbuster deal worth tens of billions of dollars.
Hengrui Pharma struck a USD 12.5 billion partnership with global pharmaceutical giant GlaxoSmithKline to co-develop up to 12 innovative medicines, setting a new record for China’s innovative-drug “going global” efforts.
Just one month earlier, China’s homegrown innovative drug Zelemi® (benvitimod cream) had also set a record.
The American Academy of Dermatology (AAD)—widely regarded as the global bellwether in dermatology—broke its all-time fastest revision record for the gold-standard treatment of atopic dermatitis, updating the guideline within 23 months and listing Zelemi® (benvitimod cream) in the highest tier as “strongly recommended.”
This means that, for the first time, dermatologists across 160 countries and regions are explicitly guided by the guideline to prioritize a Chinese medicine when treating eczema.
From the recent surge in transactions to Zelemi®’s pioneering exploration, Chinese pharmaceutical companies are providing new medicines for the world.
Breaking Records
A record has been broken!
In the first half of 2025, the total value of China’s innovative-drug license-out transactions reached nearly USD 66 billion—surpassing the full-year total of last year and setting a new record for China’s innovative drugs going global.
Innovative-drug BD deals have been booming, with deal values repeatedly hitting new highs.
On May 20, the upfront payment in Sansheng’s partnership with Pfizer reached USD 1.25 billion, with a total deal value exceeding USD 6 billion—setting a new record for the upfront payment of a domestically developed innovative drug.
On July 28, Hengrui Pharma signed a strategic cooperation agreement with GlaxoSmithKline to co-develop up to 12 innovative medicines, with an upfront payment of USD 500 million and a total potential value of up to USD 12 billion.

On October 22, Innovent Biologics reached a collaboration with Takeda on next-generation IO and ADC therapies, with an upfront payment of USD 1.2 billion and a total deal value of up to USD 11.4 billion.
BD stands for Business Development, referring to commercial activities through which pharmaceutical companies integrate resources, replenish pipelines, expand markets, or share risks via collaborations, licensing, M&A, technology transfers, and related approaches.
Licensing/out-licensing of innovative drugs is one of the most common forms of BD.
In the past, Chinese pharmaceutical companies mainly “brought in” medicines—purchasing products from multinational drugmakers.
Today, Chinese pharmaceutical companies are “going out” and expanding into international markets.
China’s innovative drugs are delivering more and better treatment options to the world.
Amid this wave, one Chinese innovative drug targeting dermatologic diseases has rewritten the treatment dilemma for hundreds of millions of patients.
Atopic dermatitis, commonly known as “eczema,” affects nearly 400 million people worldwide and is the largest disease category in dermatology.
Notably, about 10%–20% of children worldwide suffer from eczema.
Data show that the prevalence among infants under 1 year old in China is 30%, and the prevalence among children aged 1 to 7 is 13%.
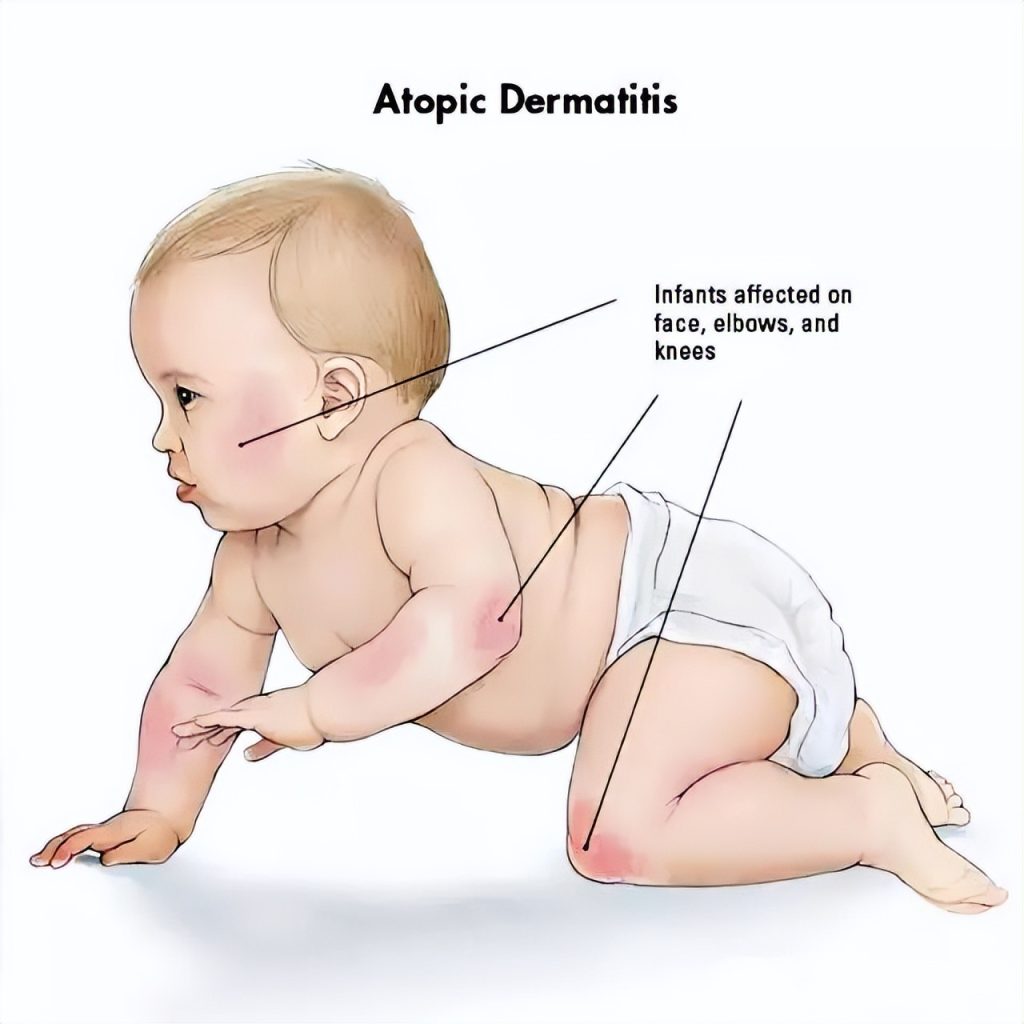
Although eczema is not fatal, it torments patients with recurrent, intense itching. In severe cases, it can even affect growth and development in children under 12, leading to insomnia, changes in appearance, and other issues—substantially reducing quality of life.
For a long time, eczema treatment has been trapped in a “double bind.”
More than 90% of patients use topical therapies. One option is corticosteroids, which act quickly but carry risks such as skin atrophy and hyperpigmentation with long-term use. Another option is topical calcineurin inhibitors, which have a favorable safety profile but tend to have a slower onset and weaker efficacy.
Children have a fragile skin barrier and are prone to relapse. Coupled with caregivers’ concerns about side effects, adherence is often poor—creating a cycle in which “without medication the child suffers, but with medication there are lingering worries.”
The range of topical options for eczema is very limited and does not adequately meet pediatric treatment needs.
It was not until 2024 that patients worldwide finally saw a new medicine.
On November 22, 2024, Zelemi® (benvitimod cream), independently developed by Shanghai Zedeman Pharmaceutical Technology Co., Ltd., was approved for marketing by China’s National Medical Products Administration (NMPA).

Just 20 days later, Zelemi® (benvitimod cream) was also approved by the U.S. FDA, achieving a simultaneous first launch in China and the United States.
Zelemi® (benvitimod cream) is a homegrown innovative drug with a brand-new mechanism of action and target. It is also the world’s first aryl hydrocarbon receptor (AhR) modulator approved for the treatment of eczema (atopic dermatitis) in children aged 2 years and older as well as adults.
As a non-steroidal topical therapy, clinical results for Zelemi® (benvitimod cream) show that itching can stop as early as the day after treatment begins. One year after discontinuation, 70% of patients did not relapse. Systemic exposure risk is extremely low, and the main adverse reaction is only mild folliculitis.
Achieving rapid onset, strong efficacy, and durable benefit simultaneously among non-steroidal therapies, this outstanding therapeutic performance drew the attention of the American Academy of Dermatology (AAD).
The AAD’s guideline updates are known for rigor: after the 2014 edition was released, it took nine years to complete the revision in 2023.
Tapinarof is the overseas name for benvitimod.
After confirming the clinical value of Zelemi® (benvitimod cream), the AAD completed an update in the shortest cycle on record and issued a “strong recommendation.”
To date, Zelemi® (benvitimod cream) has been approved for marketing in multiple countries, including China, the United States, Japan, Canada, Germany, and France.
China’s innovative drugs are bringing new therapies and new choices to hundreds of millions of patients worldwide.
A 30-Year Marathon
Innovative drug R&D has long been described as a “high-risk adventure with slim odds”—difficult, risky, time-consuming, and highly specialized.
From early research to final launch, an innovative drug typically takes 10–15 years on average, requires an investment of USD 2–3 billion, and has a clinical-stage success rate of less than 10%.
The birth of Zelemi® (benvitimod cream) is precisely the story of nearly 30 years of perseverance and breakthroughs in this “adventure.”
In the 1990s, Chen Genghui, who was pursuing a PhD in biology at Simon Fraser University in Canada, isolated—together with his team members—a stilbene-like small-molecule compound from metabolites of a symbiotic bacterium associated with a soil nematode.

This compound later became benvitimod (English name: Tapinarof), which would go on to make waves across the global dermatology field.
Although benvitimod has a molecular weight of only 254 Da, it can precisely modulate the aryl hydrocarbon receptor (AhR).
The aryl hydrocarbon receptor (AhR) is an intracellular, ligand-activated transcription factor widely expressed in skin keratinocytes, immune cells, and epithelial cells.
If the human immune system is a house, the AhR is the master sensor that can control all the lights.
Once activated, it triggers a cascade of immune responses.
Chen Genghui and his team members keenly realized that activating and modulating this “super target” via benvitimod could regulate cells involved in skin inflammation, thereby suppressing inflammation and oxidative stress and promoting restoration of skin barrier function.
Even more importantly, benvitimod also exhibits good skin permeability.
Based on its biological properties, benvitimod is well suited to be formulated as a topical medicine for the treatment of skin diseases.
In 1999, Chen Genghui, together with his classmate Li Jianxiong and their mentor Webster, formally founded a company in Canada, obtained the patent grant for benvitimod, and embarked on a systematic R&D journey.
Later, Chen Genghui returned to China to continue advancing benvitimod’s preclinical research and subsequent clinical trials.
In May 2019, benvitimod cream was approved for marketing for the treatment of mild-to-moderate plaque psoriasis in adults.
In 2021, Chen Genghui founded Zedeman Pharma in Shanghai’s Pudong New Area, embarking on his “third entrepreneurship” and launching the “second development” of benvitimod.

This time, Chen Genghui aimed to develop an AhR modulator based on benvitimod cream to tackle atopic dermatitis (eczema), the largest disease category in dermatology.
In November 2024, Zelemi® (benvitimod cream) achieved “dual submission and dual approval” in China and the United States, marking a milestone in China’s new-drug globalization.
The R&D characteristics of “high investment, high risk, high failure rate, and long cycle” determine that innovative drugs generally carry relatively high prices in their early stages.
However, Zelemi® (benvitimod cream) adhered to an affordable, patient-friendly pricing strategy, with its initial domestic price set at only one-tenth of the U.S. price.
To further improve access, Zedeman Pharma continued to optimize manufacturing efficiency, control costs, and upgrade its management system, cutting the price by 60% in July 2025.
At the same time, Zedeman Pharma also proactively participated in medical-insurance negotiations and passed the initial review for inclusion in the national reimbursement program.
Looking ahead, as policy dividends such as reimbursement inclusion are realized, people in China will be able to access top-tier innovative medicines with internationally comparable quality and efficacy at more affordable prices.
One molecule, two world-class breakthroughs!
Chen Genghui and his team have upheld innovation for 30 years—achieving success across two indications for a single drug and establishing a benchmark for China’s innovative drugs, from first launch to global recognition.
A Methodology for Innovation
From a compound in the laboratory to an innovative drug in the global spotlight, Zelemi’s innovation journey offers a replicable “methodology of innovation” for China’s new-drug R&D.
A needs-driven approach is innovation’s “compass.”
At the core of innovation is responding to real clinical needs.
Zelemi’s success stems from a precise understanding of the therapeutic dilemma in pediatric atopic dermatitis.
“Innovation can’t be innovation for its own sake—you have to first clarify where the real need lies.”
Chen Genghui admitted that his own child once suffered from eczema, and he personally witnessed the urgent clinical demand for medications suitable for children with atopic dermatitis.
This empathy and insight—rooted in lived experience—strengthened Chen Genghui’s determination to pursue the R&D direction.
This offers a lesson for Chinese pharmaceutical companies: innovation should be grounded in frontline clinical practice, anchored in patient pain points, and driven first and foremost by real needs.
Patent strategy is innovation’s “protective shield.”
For the new indication of atopic dermatitis, Zelemi built a full-chain patent matrix—from core compound patents to formulation processes, manufacturing workflows, and indication expansion—forming a tightly woven IP protection network.
This not only ensures that innovative are protected, enabling sustained investment in R&D, but also ultimately benefits more patients worldwide.
In the innovative-drug arena, the depth and breadth of patent directly determine a company’s core competitiveness.
Long-termism is innovation’s “star chart.”
From isolating the compound in the 1990s to Zelemi’s launch in 2024, Chen Genghui and his team’s innovation journey spanned nearly 30 years—through funding shortages and team turbulence—yet they consistently core technology R&D.
This resolve to “forge a sword over ten years” is precisely the quality China’s new-drug R&D needs most today.
There are no “shortcuts” in innovative-drug R&D—only steadfast commitment and long-term investment can nurture heavyweight that withstand clinical and market validation.
An ecosystem is innovation’s “accelerator.”
Although benvitimod was discovered in Canada, China achieved its complete translation from the laboratory to clinical application.
Zelemi’s success is inseparable from comprehensive support across China’s biopharmaceutical industry ecosystem.
One telling detail: Zelemi’s marketing application was accepted in China in December 2023, included in priority review in January 2024, and approved for marketing in November of the same year.
From clinical development to approval, China took only 523 days, while the United States took 1,198 days—China was more than twice as fast.
Behind this are the policy dividends unleashed by China’s drug review and approval reforms, as well as coordinated efforts enabled by a mature industry ecosystem.
Take Shanghai’s Pudong New Area—home to Zedeman Pharma—as an example: its biopharmaceutical industry has reached RMB 410 billion,more than 4,000 biopharma enterprises, with 718 innovative-drug pipelines under development—about one-fifth of the national total and 6% globally. The cumulative number of approved domestically developed innovative drugs and Class III innovative medical devices accounts for 13% and 11% of the national total, respectively.

From basic research and incubation/translation, to clinical trials, review and approval, and then to manufacturing deployment and, the “full-chain innovation ecosystem” built in Pudong New Area provided comprehensive support for Zelemi’s R&D and launch—spanning talent, capital, technology, and policy.
Zelemi’s success is the result of Zedeman Pharma’s on a needs-driven approach and deep cultivation of core technologies; it is also an inevitability stemming from China’s increasingly biopharma ecosystem and the coordinated force of policy and the market.
This provides a highly valuable practical model for China’s new-drug R&D and signals that China’s innovative drugs are moving from “single-point breakthroughs” toward a new stage of “systematic rise.”
A Steady Stream of Breakthroughs
Zelemi’s innovation legend is a vivid microcosm of China’s rise in new-drug development.
From 2015 to 2025, the number of drugs under development in China surged from 851 to 7,041 over a decade, representing a compound annual growth rate of 23.5%—3.4 times the global average (6.9%).
Even more noteworthy, China’s first-in-class drugs surged from 9 in 2015 to 120 in 2024, with its global share rising from 9% to 31.3%—ranking second worldwide and rapidly closing in on the United States’ leading position.
As Dr. Chen Genghui, inventor of benvitimod and Chairman of Shanghai Zedeman Pharmaceutical Technology Co., Ltd., put it: In the past, Chinese people waited for Western new drugs; today, the world is waiting for Chinese new drugs.
This assessment has been fully validated by the market: in the first half of 2025, the total value of China’s innovative-drug license-out transactions reached nearly USD 66 billion, surpassing last year’s full-year total.
Multinational pharmaceutical giants such as Pfizer, GlaxoSmithKline, and Takeda are spending heavily to secure China’s innovative assets, voting with real money on China’s original R&D strength.
The profound transition from “follow-on innovation” to “source innovation” is propelling China’s pharmaceutical innovation into a golden era of rising quality and efficiency—and will reshape the global balance of “voice” in healthcare.
Looking ahead, more innovative drugs like Zelemi—addressing unmet clinical needs and setting global treatment directions—will continue to emerge.
The Chinese new drugs the world is waiting for are accelerating toward arrival.

