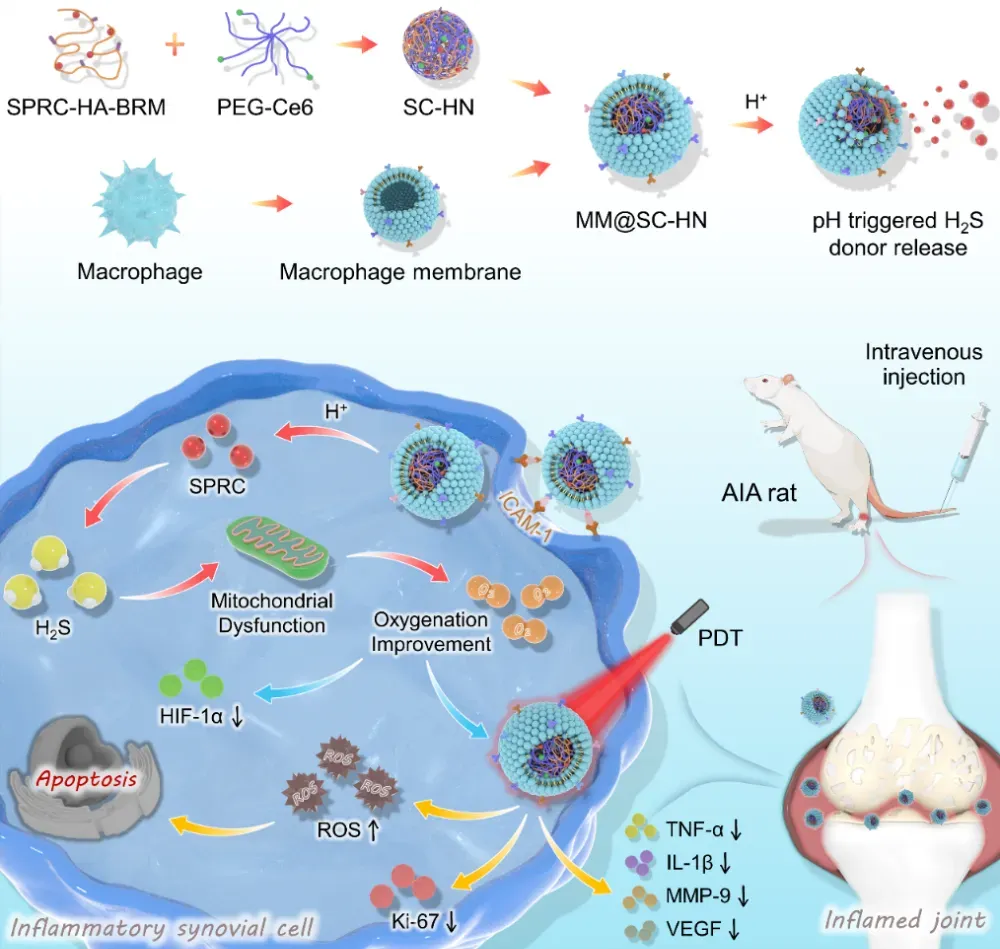
Macao University of Science and Technology (MUST) team led by Academician Zhu Yizhun, Associate Professor Guo Hui, and Associate Professor Wang Xiaolin publishes in Biomaterials: biomimetic nanoplatform overcomes intra-articular hypoxia to potentiate photodynamic therapy for rheumatoid arthritis
Rheumatoid arthritis (RA) is a complex autoimmune disease with an insidious course that can progressively erode joints, leading to swelling, pain, deformity, and even disability, affecting nearly 1% of the global population. At present, clinical management of RA mainly relies on antirheumatic drugs, nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, and biologics. Although these therapies can control disease activity to a certain extent, response rates remain suboptimal and they are often accompanied by adverse effects such as hepatotoxicity, infection, and immunosuppression. Photodynamic therapy (PDT), as a non-invasive treatment modality, generates reactive oxygen species (ROS) at lesion sites under specific light irradiation to eliminate pathological cells, offering a potentially safer, more precise, and more effective strategy for RA. However, the hypoxic environment within the joint cavity and the nonspecific distribution of photosensitizers severely limit its clinical translation.
To address these challenges, the team led by Academician Zhu Yizhun, Associate Professor Guo Hui, and Associate Professor Wang Xiaolin at Macau University of Science and Technology has, for the first time, developed a novel biomimetic nanoplatform (MM@SC-HN). This platform is cloaked with a macrophage membrane and co-loads the endogenous H₂S donor S-propargyl-cysteine (SPRC) and the photosensitizer chlorin e6 (Ce6), aiming to markedly enhance PDT efficacy by using H₂S to modulate oxygen metabolism in RA lesions. The work, entitled “Biomimetic and microenvironment-adaptive nanoplatform potentiates photodynamic therapy of rheumatoid arthritis via H2S-modulated oxygen metabolism,” was recently published in the leading materials science journal Biomaterials。

In this study, the team first loaded SPRC onto hyaluronic acid (HA), and then chemically crosslinked it with PEGylated Ce6 via an acid-sensitive small molecule to form SC-HN nanoparticles. Finally, the nanoparticles were coated with a macrophage membrane to obtain the biomimetic nanomedicine MM@SC-HN (Figure 1). In an adjuvant-induced arthritis (AIA) rat model, MM@SC-HN leveraged macrophage membrane–mediated inflammation tropism to target RA lesions. Subsequently, SPRC was released in the acidic synovial microenvironment and elevated endogenous H₂S levels to inhibit mitochondrial function, alleviate hypoxia, and enhance the Ce6-mediated photodynamic effect. Ultimately, MM@SC-HN combined with laser irradiation enabled targeted ablation of hyperplastic and inflamed synovium, remodeled the joint microenvironment, and thereby improved RA therapeutic outcomes (Figure 1).
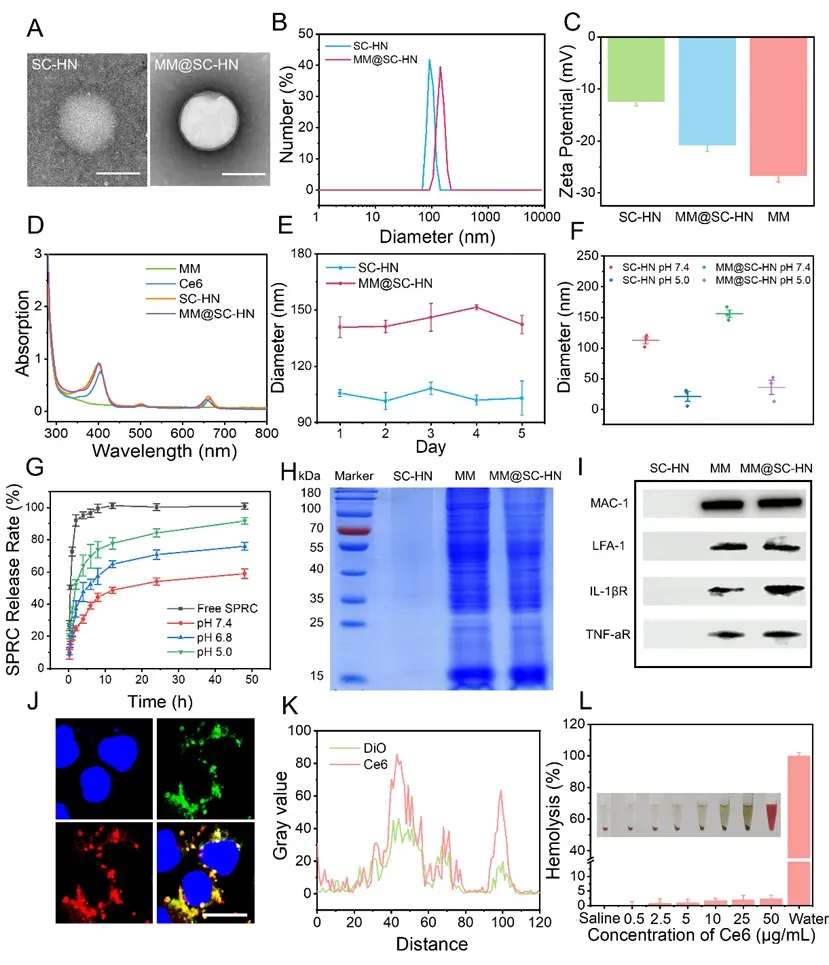
The research team then performed fundamental physicochemical characterization of MM@SC-HN. Transmission electron microscopy (TEM) images showed that MM@SC-HN had a spherical nanostructure (Figure 2A). Owing to the macrophage membrane (MM) coating, MM@SC-HN exhibited a typical core–shell morphology. Dynamic light scattering results further confirmed successful membrane coating (Figure 2B–C). MM@SC-HN showed good stability under neutral conditions, while rapidly disassembling under acidic conditions, demonstrating pH responsiveness (Figure 2E–G). In addition, MM@SC-HN displayed a protein expression profile consistent with that of the macrophage membrane, retaining key functional proteins such as lymphocyte function–associated antigen 1 (LFA-1) and receptors for inflammatory cytokines (Figure 2H–I), which are critical for inflammation chemotaxis within the joint microenvironment. Colocalization experiments under confocal microscopy verified that MM@SC-HN maintained good structural integrity during cellular transport (Figure 2J–K). Moreover, an in vitro hemolysis assay preliminarily demonstrated favorable blood compatibility of this nanoplatform, meeting the basic biosafety requirements for intravenous administration (Figure 2L).
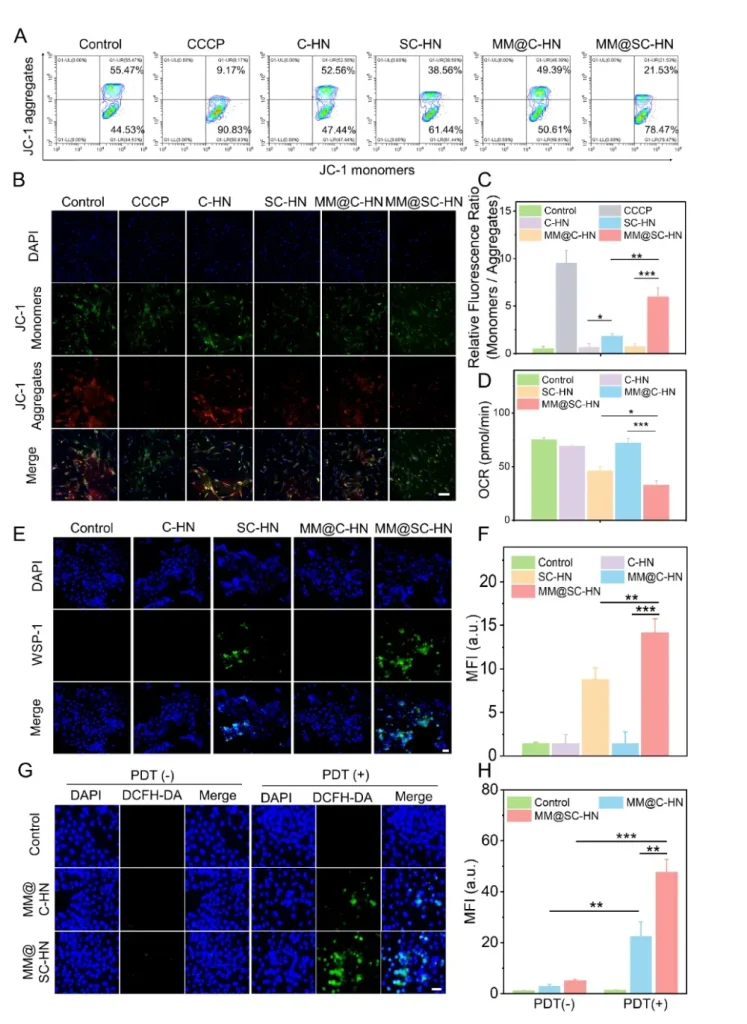
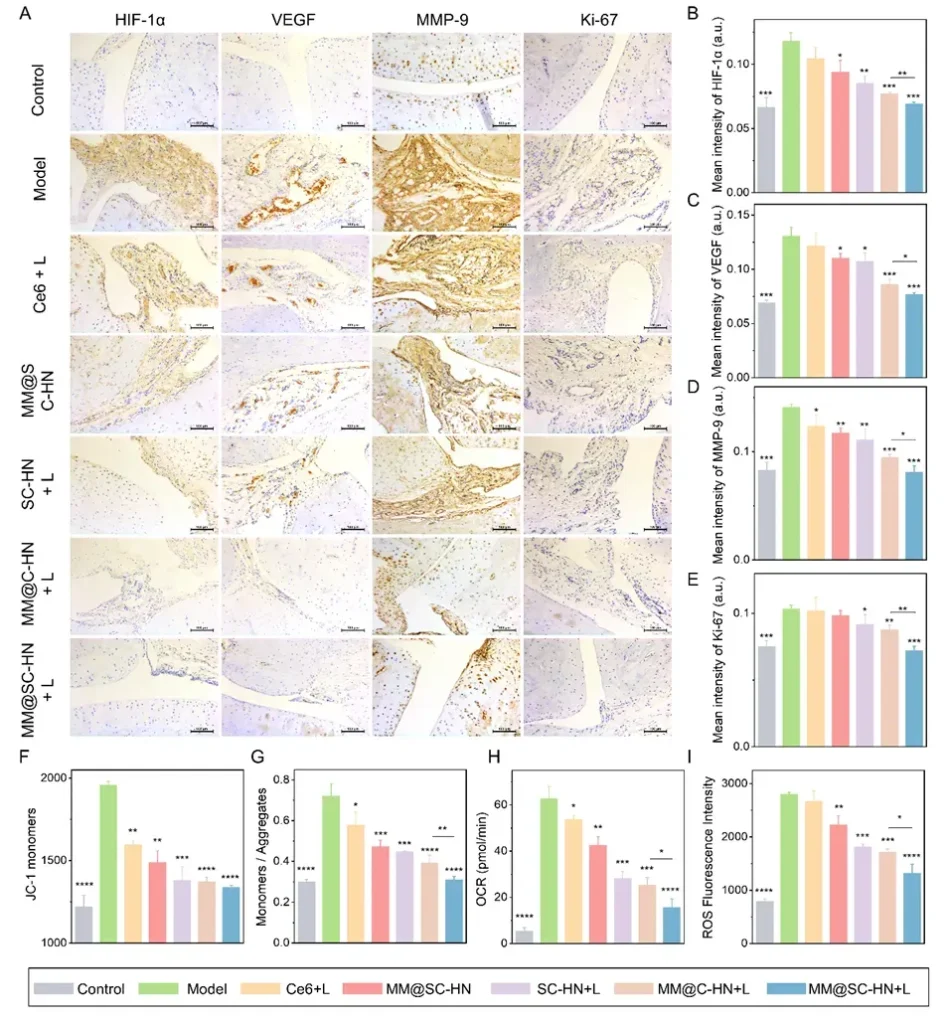
At the cellular level, fluorescence imaging and flow cytometry analyses showed that MM@SC-HN effectively delivered SPRC and generated endogenous H2S to suppress mitochondrial function, reduce cellular oxygen consumption rate (OCR), improve oxygen metabolism, and thereby enhance the photodynamic effect (Figure 3).
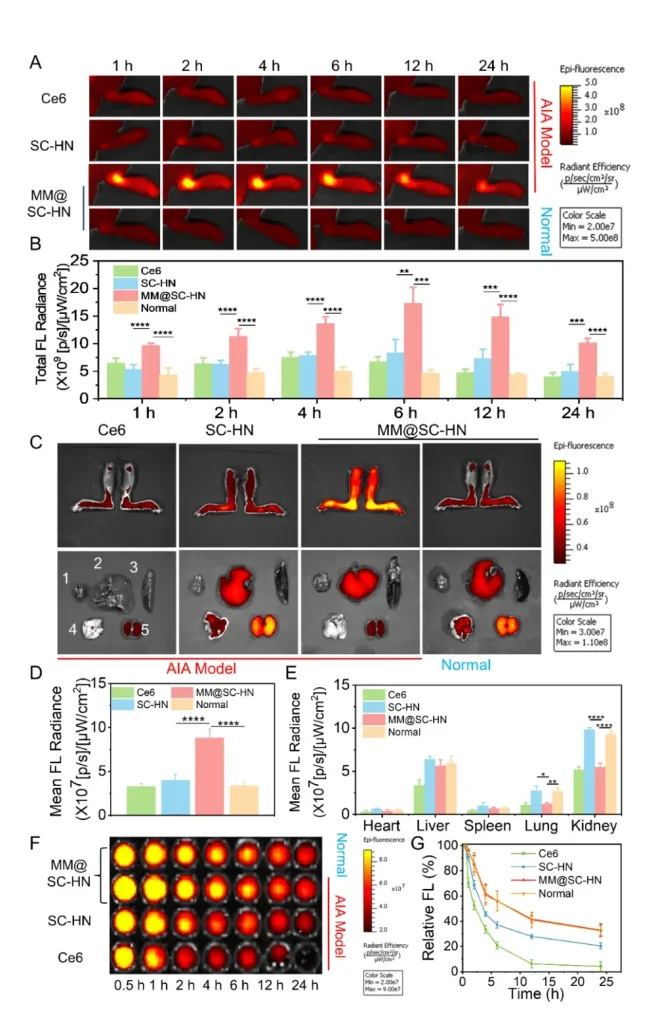
Next, leveraging the near-infrared imaging properties of Ce6, the team monitored the in vivo biodistribution and metabolic behavior of MM@SC-HN using a small-animal in vivo imaging system. Benefiting from the inflammation-homing capability of the macrophage membrane, MM@SC-HN exhibited excellent targeting to inflamed joints in vivo (Figure 4). In addition, MM@SC-HN demonstrated outstanding joint targeting and long-circulation performance. Therefore, MM@SC-HN is expected to enable integrated diagnosis and therapy for rheumatoid arthritis.
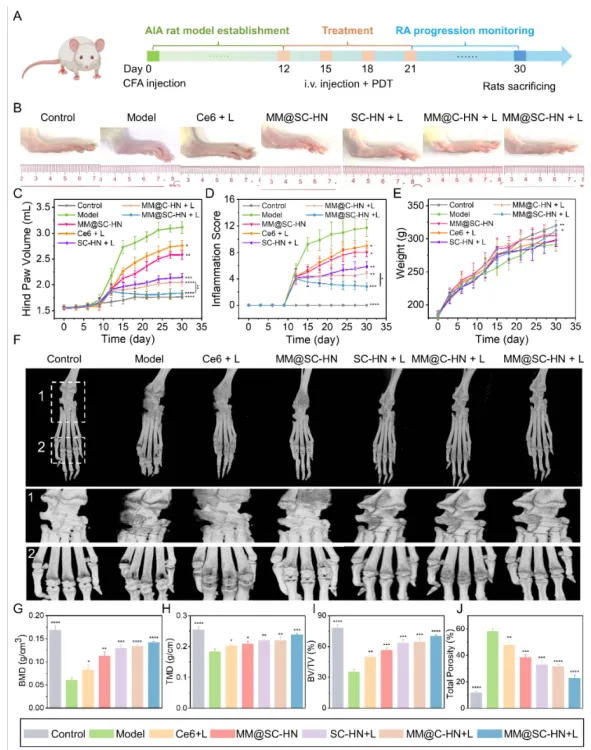
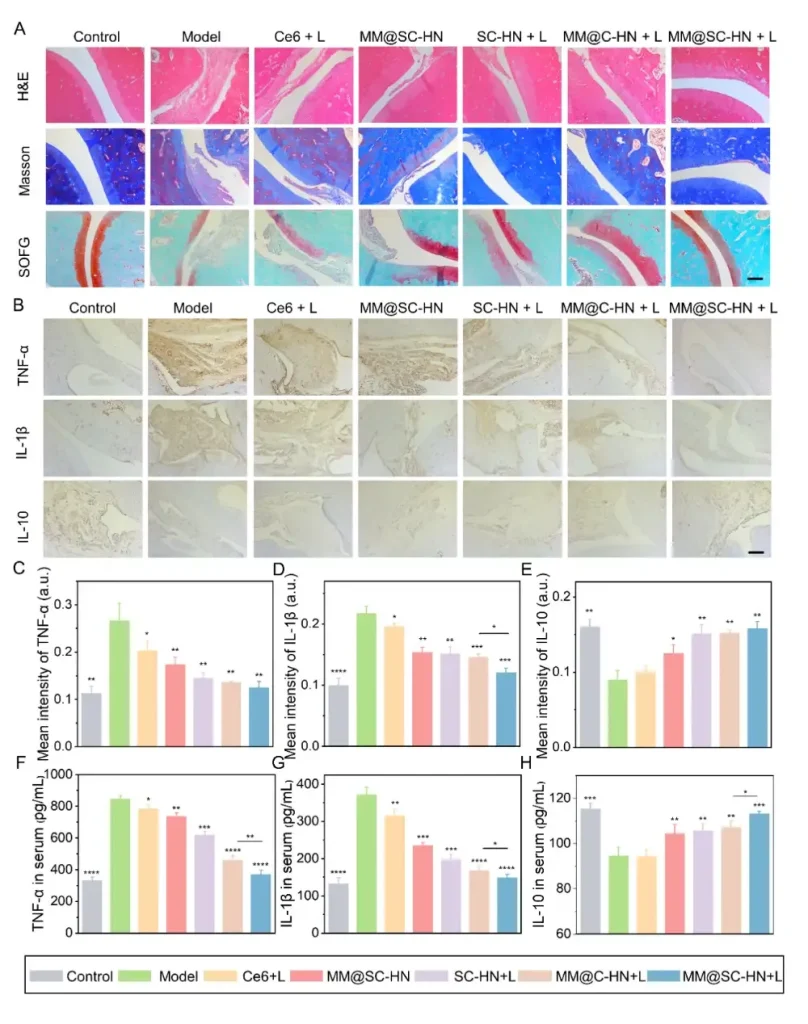
In the AIA rat model, the research team systematically evaluated the overall therapeutic efficacy of MM@SC-HN (Figures 5–7). The results showed that MM@SC-HN therapy reshaped the complex arthritic joint microenvironment across multiple key pathological processes: it not only significantly alleviated local inflammation and hypoxia, hindered pathological pannus formation and abnormal synovial hyperplasia, but also effectively corrected oxidative stress imbalance. This systemic regulation of the microenvironment translated into robust joint protection and repair: treated rats exhibited a significant and sustained reduction in paw swelling, markedly decreased histological inflammation scores, effective suppression of cartilage destruction, substantially reduced bone erosion, and signs of active subchondral bone repair. These data strongly demonstrate that, by intelligently modulating the joint microenvironment, MM@SC-HN successfully redirected disease progression from “destruction” toward “repair,” providing a new nano-therapeutic strategy for targeted intervention in rheumatoid arthritis.
In summary, this study constructed a multifaceted biomimetic nanosystem, MM@SC-HN, which ingeniously integrates a macrophage membrane coating with SC-HN nanoparticles co-loaded with the H₂S donor SPRC and the photosensitizer Ce6. The combination of hydrogen sulfide gas and photodynamic therapy effectively reversed the pathological progression of RA joints. This work not only provides a candidate nanomedicine for RA clinical treatment, but also offers a generalizable new strategy to enhance PDT efficacy in a broad range of inflammatory diseases.
The first author of this paper is PhD student Zhang Xican at Macau University of Science and Technology, and the corresponding authors are Academician Zhu Yizhun, Associate Professor Guo Hui, and Associate Professor Wang Xiaolin. This work was supported by the National Natural Science Foundation of China, the Macao Science and Technology Development Fund, the Natural Science Foundation of Guangdong Province, and the Shenzhen Science and Technology Innovation Commission Guangdong–Hong Kong–Macao Greater Bay Area Class C Project, among others. The research group is well funded and is recruiting postdoctoral fellows as well as master’s and doctoral students on a long-term basis. Interested applicants are welcome to apply (xilwang@must.edu.mo).

