2025 Academic Committee Meeting of the Laboratory of Natural Drug Discovery and Industrialization Successfully Held
The 2025 Academic Committee Meeting of the Laboratory for Natural Drug Discovery and Industrialization was successfully held recently in Meeting Room N502 at Macao University of Science and Technology (MUST). The meeting was chaired by Academician Yang Baofeng of the Chinese Academy of Engineering and renowned pharmacologist, and led by Academician Zhu Yizhun, Member of the European Academy of Sciences and Arts, Associate Vice President of MUST and Dean of the Faculty of Pharmacy, Chief Scientist of China’s National Basic Research Program (973 Program), and Director of the Laboratory for Natural Drug Discovery and Industrialization. A number of leading experts and scholars from China and abroad, as well as representatives of relevant institutions, attended the meeting in a hybrid online–offline format to jointly explore pathways for the laboratory’s high-quality development and innovation in natural drug industrialization.

Distinguished guests included Yu Yusheng, Chairman of the Administrative Committee of the Macao Science and Technology Development Fund; Chen Jimin, Vice Chairman of the University Council and Secretary-General of MUST; Professor Du Zhimin of Harbin Medical University; Professor Zhu Yichun, Professor and Head of the Department of Physiology and Pathophysiology at Fudan University; Zhou Demin, Director of the Ningbo Institute of Ocean Medicine at Peking University and Director of the State Key Laboratory of Natural Products and Biomimetic Drugs; Zeng Fanyi, Director of the Institute of Medical Genetics at Shanghai Jiao Tong University; Ma Jing, Director of the Shanghai Center for Safety Evaluation of New Drugs; Wang Mingwei, Chair Professor at Fudan University; Professor Liu Peiqing, Professor at the School of Pharmaceutical Sciences, Sun Yat-sen University and Director of the Laboratory of Pharmacology and Toxicology; Huang Min, Director of the Institute of Clinical Pharmacology, Sun Yat-sen University; and Feng Qizhu, Senior Technical Officer of the Administrative Committee of the Macao Science and Technology Development Fund.

At the beginning of the meeting, Chairman Yu Yusheng delivered opening remarks on behalf of the Macao Science and Technology Development Fund. He noted that since its launch in 2022, the Laboratory for Natural Drug Discovery and Industrialization has remained focused on core areas of natural drug R&D. The laboratory has established a 34-member high-caliber research team, trained multiple outstanding graduate students, published a number of high-quality SCI papers, and filed 15 patent applications, achieving notable progress in technology transfer and translation. The Fund has cumulatively invested MOP 19.86 million to support the laboratory’s establishment and development. In addition to advancing industrialization, the cultivation of young talent is also a key priority, as it is a critical link in accelerating the translation and commercialization of research outputs. Looking ahead, the Fund will continue to provide strong support for the laboratory to expedite new drug translation, deepen international academic and industry collaboration, and help establish the laboratory as a global hub for natural drug R&D, enabling breakthrough progress in Macao’s pharmaceutical industrialization.

In his remarks, Vice Chairman Chen Jimin stated that the Laboratory for Natural Drug Discovery and Industrialization is a core research platform for biomedicine at MUST. Under the leadership of Academician Zhu Yizhun, the team has worked in unity, tackled challenging problems, and achieved a series of landmark research outcomes. The University attaches great importance to the laboratory’s development, providing more than MOP 30 million in cumulative matching support, including high-quality facilities and advanced research equipment, thereby laying a solid foundation for the laboratory’s work. The laboratory has a well-structured team with a robust talent pipeline, and it has established in-depth collaborations with well-known enterprises such as Beijing Tong Ren Tang, effectively promoting deep integration between research and industry. Going forward, the University will continue to provide comprehensive support in talent recruitment and development, platform development, and resource allocation, enabling the laboratory to further expand external collaboration, accelerate the translation of research outputs, and contribute more to the high-quality development of Macao’s health industry.

Subsequently, Laboratory Director Academician Zhu Yizhun delivered a detailed report on the laboratory’s key progress and major accomplishments in 2025. He explained that the laboratory has further improved and strengthened its infrastructure and research team, and is currently focused on four core R&D pipelines: (1) research and development of SPRC derived from natural products for the treatment of rheumatoid arthritis; (2) research and development on the druggability of rhynchophylline, a natural product, for glycemic control and hepatoprotection; (3) development of small-molecule SMYD3 inhibitors from natural products aimed at delaying vascular aging; and (4) pre-NDA studies of SCM-198, a first-in-class natural product–derived innovative drug candidate based on leonurine. Each pipeline has achieved major milestone progress.
Among these, SPRC was approved in July 2025 to initiate a Phase I clinical trial for the treatment of rheumatoid arthritis. The medicinal crystalline form of leonurine has successfully obtained clinical trial authorization as a Category 1 original chemical drug, and a Phase I clinical study involving 106 subjects has been completed. The results indicate favorable safety and tolerability, and the program has now progressed smoothly into Phase II. In parallel, the laboratory’s research achievements have received multiple honors, including the Second Prize of the 2024 Macao Technological Invention Award and the Gold Award for High-Value Patents in the Guangdong–Hong Kong–Macao Greater Bay Area. The laboratory has also secured 38 authorized invention patents in China, as well as 16 invention patents for new drugs in the United States and internationally (PCT), and in Japan. Over the past year, the laboratory obtained MOP 55.86 million in new competitive research funding and published 21 SCI papers in internationally recognized academic journals, further strengthening its research capabilities and academic influence.
Academician Zhu Yizhun stated that the laboratory will further leverage the top-tier scientific resources of MUST and Peking University, focusing on key challenges in upstream discovery for natural and innovative drugs, and prioritizing projects such as drug delivery and pre-market research for four original innovative drug candidates. Looking ahead, the laboratory plans to apply for designation as a National Engineering Center, supporting Macao’s pharmaceutical industry in transitioning from an “integration of generics and innovation” model to an “originating innovation” model, breaking through regional development bottlenecks and filling a regional gap in establishing a national-level industrialization incubation platform in the field of natural drugs. The experts attending the Academic Committee highly affirmed the laboratory’s outstanding achievements in basic research and clinical translation, and provided a range of constructive recommendations and guidance on key topics including future development pathways, scaling strategy, annual planning, and the National Engineering Center application.

Finally, Academic Committee Chair Academician Yang Baofeng delivered concluding remarks, fully acknowledging the laboratory’s achievements in establishment and development, and expressing strong expectations and best wishes for its continued innovation and growth. Academician Zhu Yizhun thanked the experts for their guidance and recommendations, and stated that the laboratory will carefully review and incorporate the constructive feedback, further strengthen research and translational efforts in the coming year, fully advance the National Engineering Center application, accelerate clinical translation of products, and make greater contributions to the high-quality development of Macao’s biomedical industry and the coordinated development of the Greater Bay Area’s health industry.
Top-Tier Journal Research: Garlic Outperforms Metformin in Longevity Extension
On December 19, 2025, the University of Seville in Spain published a research paper in the journal Cell Metabolism.


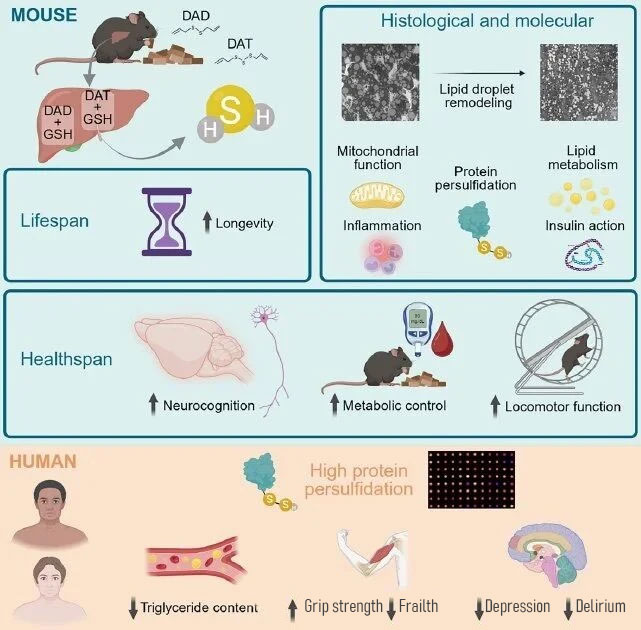
This study is the first to demonstrate in mammals that diallyl sulfide (H2S) from garlic can continuously generate hydrogen sulfide through non-enzymatic pathways, extending mouse lifespan by 11.4%—a greater extension than metformin (5.8%)—while improving metabolic, motor, and cognitive functions.
The research team also analyzed human data, finding that among patients with multiple comorbidities, higher plasma protein persulfidation levels were associated with stronger grip strength and lower triglyceride levels, further supporting the potential role of H2S in maintaining human health.
The researchers stated that this study is the first to confirm in mammals that natural compounds from garlic can generate H2S through non-enzymatic pathways, thereby extending lifespan and improving health status. Diallyl sulfide from garlic is safe and readily accessible, providing scientific evidence for developing dietary intervention-based anti-aging strategies.
In Less Than a Month, Marketing Applications for Over 100 Generic Drugs Rejected

Recently, the National Medical Products Administration (NMPA) has been frequently publishing delivery notices for drug notification letters on its official website. Generally speaking, delivery of such a notification letter indicates that a marketing authorization application has been “not approved,” or that the applicant has proactively withdrawn the application after anticipating it would not pass review. Based on statistics compiled by Yicai (First Financial) from the NMPA website, from the beginning of December through December 19, the list involved 167 drugs, of which 102 were chemical generic drug applications (based on acceptance numbers starting with CYHS). In other words, in December, 102 chemical generic drug marketing applications were either rejected or withdrawn—double the number in November. What signal does this send?
These Generic Drug Applications Are Running Into Headwinds
Since December, which specific chemical generic drug applications among these 102 were rejected or voluntarily withdrawn?
By therapeutic area, the applications span cardiovascular and metabolic diseases, the nervous and respiratory systems, anti-infectives, oncology, ophthalmology, dermatology, gastroenterology, and more. The drugs involved include sacubitril/valsartan sodium tablets, sitagliptin/metformin extended-release tablets, vortioxetine hydrobromide tablets, ciclesonide inhalation aerosol, levofloxacin tablets, etc. For some products, multiple manufacturers encountered difficulties with their applications. The allergy drug bilastine involved the largest number of companies—six in total—including Shandong Langnuo Pharmaceutical, Yangzijiang Pharmaceutical Group Beijing Haiyan Pharmaceutical, Jiangsu Hechen Pharmaceutical, among others, covering oral solution, tablets, and orally disintegrating tablets. Following bilastine, three companies were involved for both the brexpiprazole product line for psychiatric disorders and the levofloxacin antibiotic series; the antihypertensive levamlodipine besylate tablets involved two companies. In a few cases, listed companies issued announcements to explain the situation. For example, Jiudian Pharmaceutical’s anti-inflammatory and analgesic indomethacin gel patch was also on the delivery list. The company stated that additional trial data were required; after communication with the Center for Drug Evaluation (CDE) of the NMPA, it decided to withdraw the registration application, and will restart the filing process after supplementing and improving the relevant studies. A generic drug manufacturer whose application was rejected told Yicai reporters that the issue this time mainly concerned the active pharmaceutical ingredient (API), and was unrelated to the company’s formulation itself.
Tighter Approval Review
Among the drugs whose applications were rejected or withdrawn this time are not a few major market products, or products that already have a large number of approved manufacturers. Take amlodipine besylate tablets, a common antihypertensive, for example: the originator drug is Norvasc, and more than 60 generic manufacturers have already obtained marketing approvals. Several industry insiders told Yicai reporters that, judging from current developments, the review and approval of chemical generic drugs is trending tighter. In their view, policy signals are also pointing in this direction. Two draft documents organized and drafted by the CDE of the NMPA—“Major Defects in Pharmaceutical Research for Chemical Generic Drugs (Trial) (Draft for Comments)” and “Major Defects in Bioequivalence Studies for Chemical Generic Drugs (Trial) (Draft for Comments)”—were released on December 3. In the former draft, for major defects, applicants would no longer be asked to provide supplementary materials; instead, a decision of non-approval would be made based on the submitted dossier. The latter draft provides detailed descriptions of major defects in bioequivalence studies for chemical generic drugs, including insufficient or inadequate bioequivalence studies, deficiencies in analytical testing, deficiencies in statistical analysis, insufficient justification for protocol deviations, and errors or omissions in the submission dossier, aiming to clarify what constitutes major defects in bioequivalence research. “At the beginning of this month, the CDE clarified the standards for generic drugs failing review, and then we saw a sizable wave of withdrawals; this appears to be a consistent follow-through of policy implementation,” said Zhou Liyun, Chairman of PharmaCube (Yiyao Mofang).
Reducing Low-Level Redundancy
China is a major producer of generic drugs. In recent years, the inclusion of more innovative drugs in the national reimbursement list and the expansion of volume-based procurement (VBP) have significantly compressed generic drug margins—measures intended to steer domestic pharmaceutical companies toward innovation. However, the growing number of generic drug companies and product filings is further intensifying competition in an already mature market. According to the China Generic Drug Development Report (2025) (the “Report”), jointly released recently by the Institute of Materia Medica of the Chinese Academy of Medical Sciences, the China Pharmaceutical Industry Information Center, and the National Institutes for Food and Drug Control, chemical generic drugs still accounted for 50% of the overall pharmaceutical market in 2024. The top 100 chemical generic drug companies by sales contributed 55% of the market, a share showing a slow downward trend. The combined market share of the top 10 chemical generic drug companies—such as CSPC Pharmaceutical Group, Qilu Pharmaceutical, and Hengrui Medicine—has remained stable at around 22%, indicating that market concentration is still relatively low. The Report also shows that in 2024, the number of generic drug varieties that passed, or were deemed to have passed, the consistency evaluation reached 914, a substantial increase from 543 in 2021. Among these, the number of varieties with five or more approved manufacturers increased from 79 in 2021 to 203, with intensified homogenization becoming the backdrop of generic drug competition. An industry source told Yicai reporters that some generic drug companies may initiate many generic projects each year, with investment of several million yuan per project; compared with the R&D costs of innovative drugs—which can exceed hundreds of millions or even billions of yuan—this cost remains relatively low. In past rounds of chemical drug VBP, some products saw aggressive bidding due to the sheer number of competing companies. Efforts to curb “involution”—excessive low-value competition—in the generic drug sector have drawn broad attention. In May this year, a study published in Chinese Journal of Modern Applied Pharmacy by authors from institutions including the National Drug Policy and Pharmaceutical Industry Economic Research Center of China Pharmaceutical University found that China’s generic drug sector faces issues such as low-level repetitive development, and recommended optimizing the review and approval mechanism to support sustained high-quality development of generics. “For products where competition is already extremely crowded, the market value of continued development is very limited. Policy should indeed provide some guidance to reduce involution and avoid unnecessary waste of industry resources,” Zhou Liyun also said.
Macao University of Science and Technology (MUST) team led by Academician Zhu Yizhun, Associate Professor Guo Hui, and Associate Professor Wang Xiaolin publishes in Biomaterials: biomimetic nanoplatform overcomes intra-articular hypoxia to potentiate photodynamic therapy for rheumatoid arthritis
Rheumatoid arthritis (RA) is a complex autoimmune disease with an insidious course that can progressively erode joints, leading to swelling, pain, deformity, and even disability, affecting nearly 1% of the global population. At present, clinical management of RA mainly relies on antirheumatic drugs, nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, and biologics. Although these therapies can control disease activity to a certain extent, response rates remain suboptimal and they are often accompanied by adverse effects such as hepatotoxicity, infection, and immunosuppression. Photodynamic therapy (PDT), as a non-invasive treatment modality, generates reactive oxygen species (ROS) at lesion sites under specific light irradiation to eliminate pathological cells, offering a potentially safer, more precise, and more effective strategy for RA. However, the hypoxic environment within the joint cavity and the nonspecific distribution of photosensitizers severely limit its clinical translation.
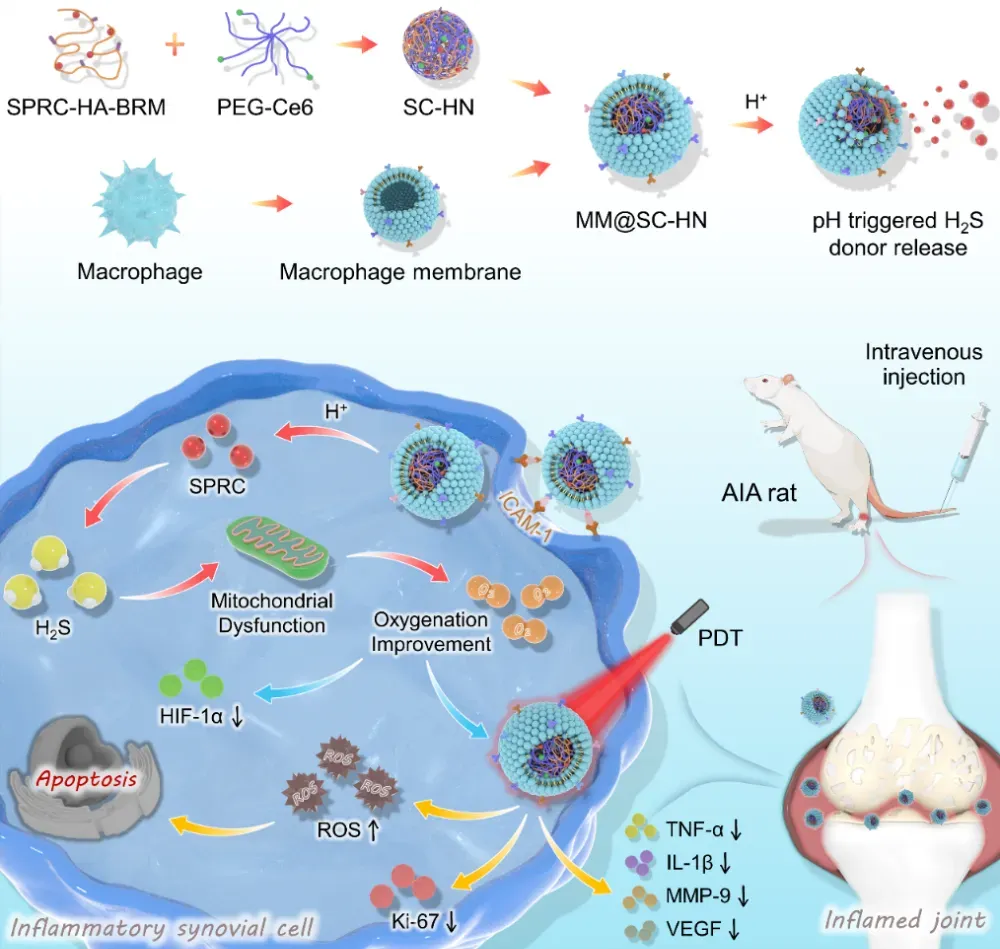
To address these challenges, the team led by Academician Zhu Yizhun, Associate Professor Guo Hui, and Associate Professor Wang Xiaolin at Macau University of Science and Technology has, for the first time, developed a novel biomimetic nanoplatform (MM@SC-HN). This platform is cloaked with a macrophage membrane and co-loads the endogenous H₂S donor S-propargyl-cysteine (SPRC) and the photosensitizer chlorin e6 (Ce6), aiming to markedly enhance PDT efficacy by using H₂S to modulate oxygen metabolism in RA lesions. The work, entitled “Biomimetic and microenvironment-adaptive nanoplatform potentiates photodynamic therapy of rheumatoid arthritis via H2S-modulated oxygen metabolism,” was recently published in the leading materials science journal Biomaterials。

In this study, the team first loaded SPRC onto hyaluronic acid (HA), and then chemically crosslinked it with PEGylated Ce6 via an acid-sensitive small molecule to form SC-HN nanoparticles. Finally, the nanoparticles were coated with a macrophage membrane to obtain the biomimetic nanomedicine MM@SC-HN (Figure 1). In an adjuvant-induced arthritis (AIA) rat model, MM@SC-HN leveraged macrophage membrane–mediated inflammation tropism to target RA lesions. Subsequently, SPRC was released in the acidic synovial microenvironment and elevated endogenous H₂S levels to inhibit mitochondrial function, alleviate hypoxia, and enhance the Ce6-mediated photodynamic effect. Ultimately, MM@SC-HN combined with laser irradiation enabled targeted ablation of hyperplastic and inflamed synovium, remodeled the joint microenvironment, and thereby improved RA therapeutic outcomes (Figure 1).
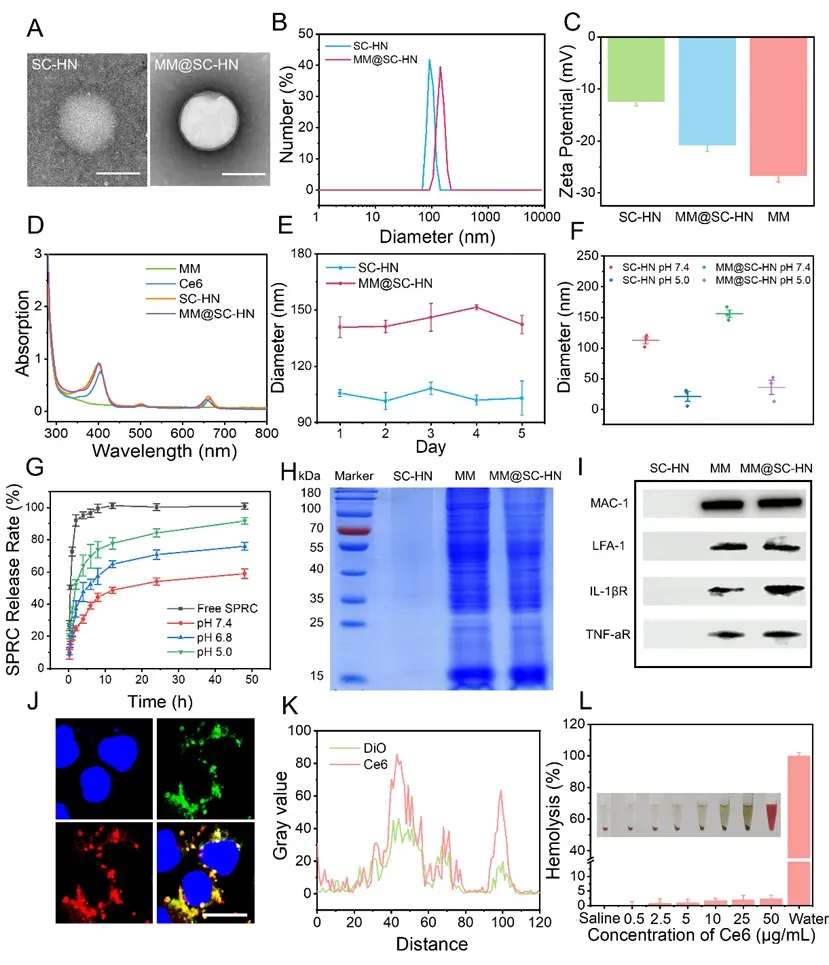
The research team then performed fundamental physicochemical characterization of MM@SC-HN. Transmission electron microscopy (TEM) images showed that MM@SC-HN had a spherical nanostructure (Figure 2A). Owing to the macrophage membrane (MM) coating, MM@SC-HN exhibited a typical core–shell morphology. Dynamic light scattering results further confirmed successful membrane coating (Figure 2B–C). MM@SC-HN showed good stability under neutral conditions, while rapidly disassembling under acidic conditions, demonstrating pH responsiveness (Figure 2E–G). In addition, MM@SC-HN displayed a protein expression profile consistent with that of the macrophage membrane, retaining key functional proteins such as lymphocyte function–associated antigen 1 (LFA-1) and receptors for inflammatory cytokines (Figure 2H–I), which are critical for inflammation chemotaxis within the joint microenvironment. Colocalization experiments under confocal microscopy verified that MM@SC-HN maintained good structural integrity during cellular transport (Figure 2J–K). Moreover, an in vitro hemolysis assay preliminarily demonstrated favorable blood compatibility of this nanoplatform, meeting the basic biosafety requirements for intravenous administration (Figure 2L).
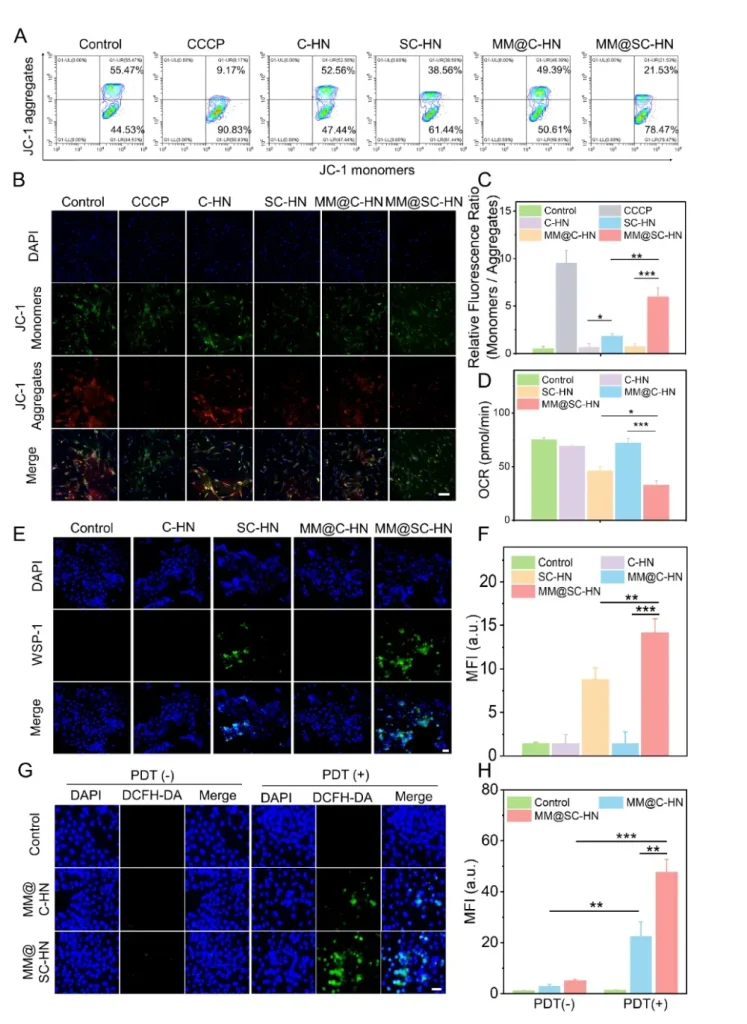
At the cellular level, fluorescence imaging and flow cytometry analyses showed that MM@SC-HN effectively delivered SPRC and generated endogenous H2S to suppress mitochondrial function, reduce cellular oxygen consumption rate (OCR), improve oxygen metabolism, and thereby enhance the photodynamic effect (Figure 3).
Next, leveraging the near-infrared imaging properties of Ce6, the team monitored the in vivo biodistribution and metabolic behavior of MM@SC-HN using a small-animal in vivo imaging system. Benefiting from the inflammation-homing capability of the macrophage membrane, MM@SC-HN exhibited excellent targeting to inflamed joints in vivo (Figure 4). In addition, MM@SC-HN demonstrated outstanding joint targeting and long-circulation performance. Therefore, MM@SC-HN is expected to enable integrated diagnosis and therapy for rheumatoid arthritis.
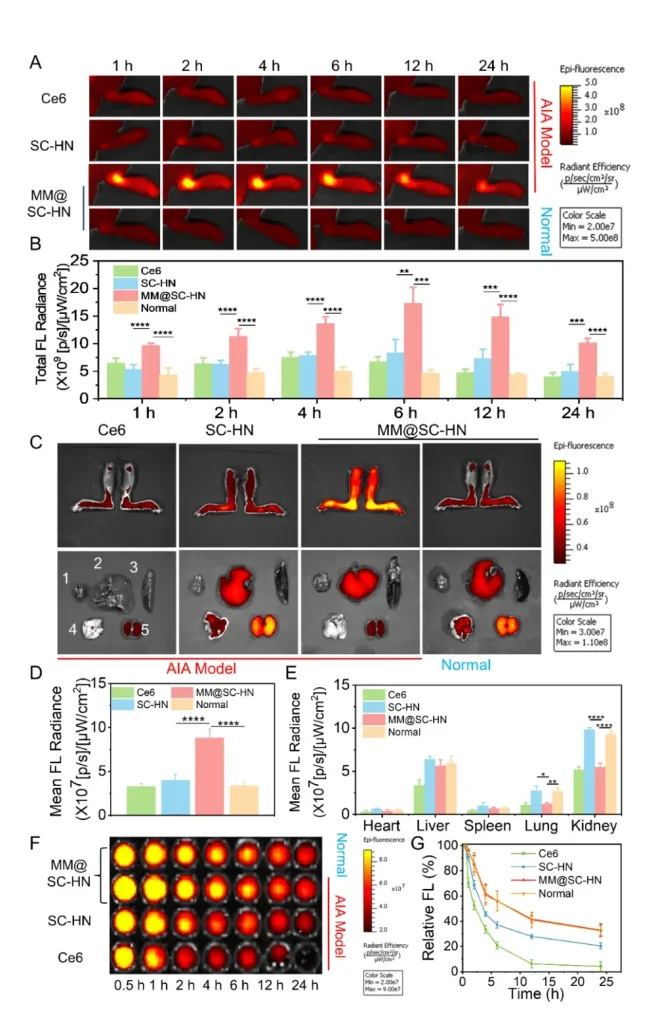
In the AIA rat model, the research team systematically evaluated the overall therapeutic efficacy of MM@SC-HN (Figures 5–7). The results showed that MM@SC-HN therapy reshaped the complex arthritic joint microenvironment across multiple key pathological processes: it not only significantly alleviated local inflammation and hypoxia, hindered pathological pannus formation and abnormal synovial hyperplasia, but also effectively corrected oxidative stress imbalance. This systemic regulation of the microenvironment translated into robust joint protection and repair: treated rats exhibited a significant and sustained reduction in paw swelling, markedly decreased histological inflammation scores, effective suppression of cartilage destruction, substantially reduced bone erosion, and signs of active subchondral bone repair. These data strongly demonstrate that, by intelligently modulating the joint microenvironment, MM@SC-HN successfully redirected disease progression from “destruction” toward “repair,” providing a new nano-therapeutic strategy for targeted intervention in rheumatoid arthritis.
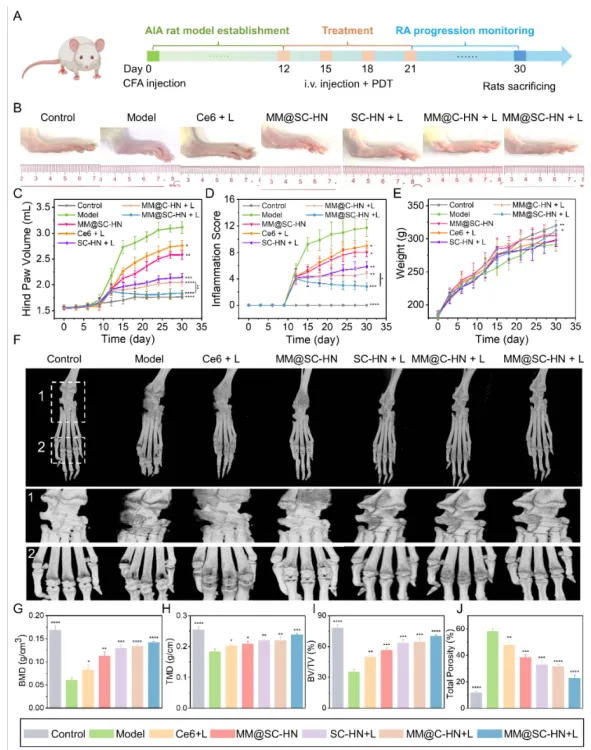
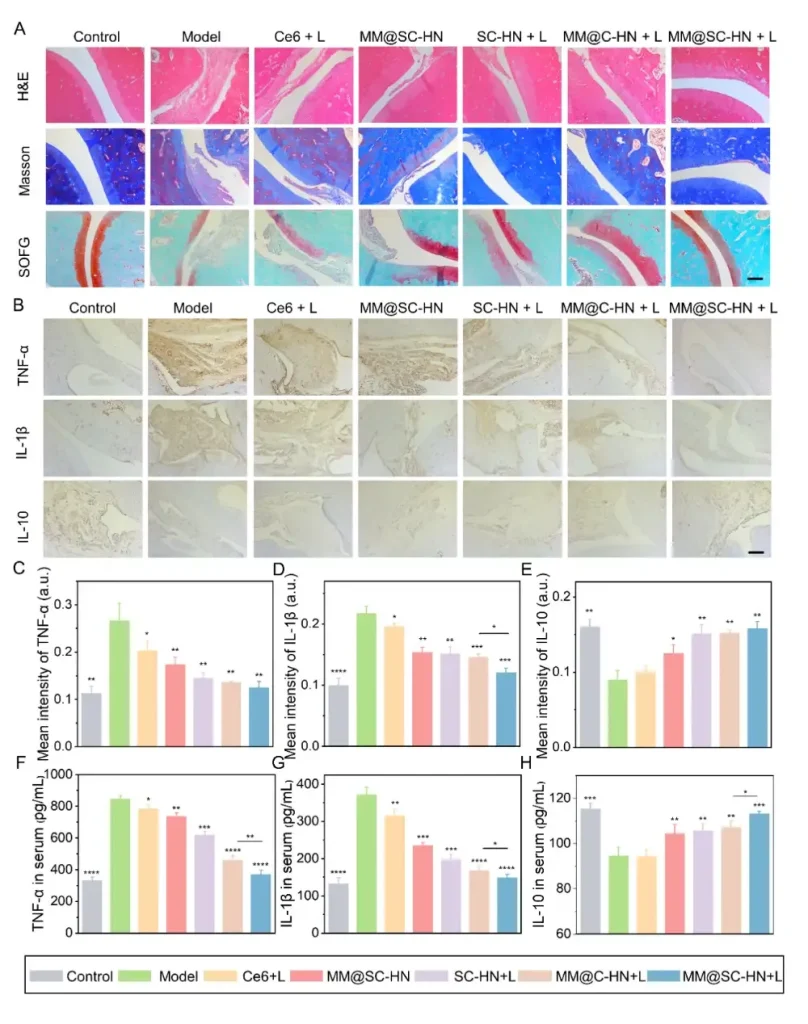
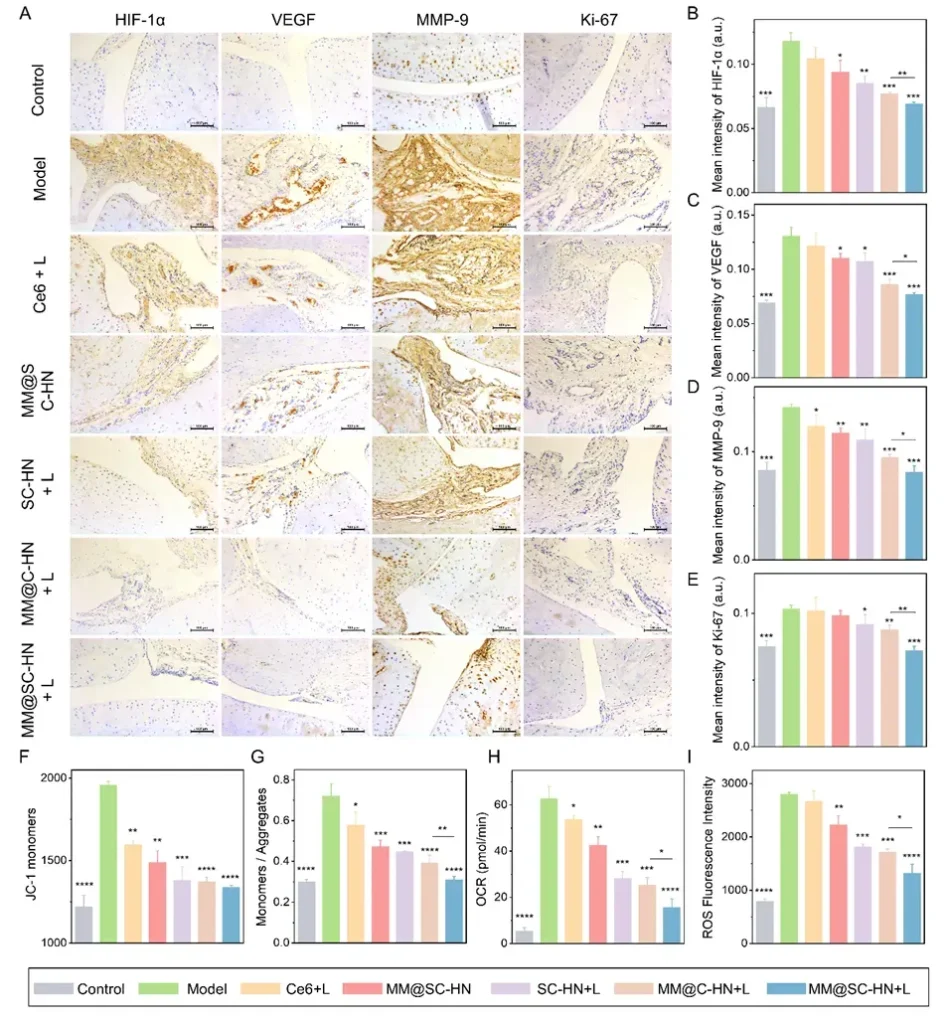
In summary, this study constructed a multifaceted biomimetic nanosystem, MM@SC-HN, which ingeniously integrates a macrophage membrane coating with SC-HN nanoparticles co-loaded with the H₂S donor SPRC and the photosensitizer Ce6. The combination of hydrogen sulfide gas and photodynamic therapy effectively reversed the pathological progression of RA joints. This work not only provides a candidate nanomedicine for RA clinical treatment, but also offers a generalizable new strategy to enhance PDT efficacy in a broad range of inflammatory diseases.
The first author of this paper is PhD student Zhang Xican at Macau University of Science and Technology, and the corresponding authors are Academician Zhu Yizhun, Associate Professor Guo Hui, and Associate Professor Wang Xiaolin. This work was supported by the National Natural Science Foundation of China, the Macao Science and Technology Development Fund, the Natural Science Foundation of Guangdong Province, and the Shenzhen Science and Technology Innovation Commission Guangdong–Hong Kong–Macao Greater Bay Area Class C Project, among others. The research group is well funded and is recruiting postdoctoral fellows as well as master’s and doctoral students on a long-term basis. Interested applicants are welcome to apply (xilwang@must.edu.mo).
China’s innovative-drug BD now accounts for nearly half of global deal flow, yet per-capita spend is only 1/124 of the U.S.: how can Chinese pharma chart a path to an Eli Lilly–scale, trillion-dollar valuation?
①The capital ecosystem in the pharmaceutical industry is undergoing a profound shift, with BD financing surpassing the primary market to become a core source of cash flow for many innovative drug companies;
②While China’s innovative drugs are strong in outbound BD and global partnering, the domestic market still lags significantly—per capita spending on innovative drugs is only 1/124 of that in the United States;
③To break the deadlock, experts recommend using commercial health insurance as a breakthrough to build a diversified payment system, while strengthening policy support and improving market inclusiveness.

Cailian Press, Dec 2 (Reporter Wu Chao) As Eli Lilly’s market capitalization recently surpassed the USD 1 trillion milestone—becoming the first company in the global biopharmaceutical industry to enter the “trillion-dollar club”—many leading figures in China’s pharmaceutical sector have been asking a question: When will China produce its own “Eli Lilly”?
At the 2025 China Pharmaceutical City Big Health Industry Forum—hosted by the Taizhou Municipal People’s Government and organized by Tongxieyi—as well as the 16th Taizhou Pharmaceutical Expo, experts presented a set of thought-provoking figures: China’s innovative drug market is roughly 3% the size of the United States, and per capita spending on innovative drugs is only one one-hundred-and-twenty-fourth of that in the U.S.
Meanwhile, another picture is emerging: China’s outbound transactions for innovative drugs now account for nearly half of global deals, and the number of clinical trials for innovative drugs has even surpassed that of the United States.
Together, these trends underscore the reality that China’s pharmaceutical industry stands at a crossroads. China’s innovation capacity has begun to reshape the global landscape and has attracted increasing attention and favor from global capital and industry; however, industrial development and domestic market potential remain mismatched, and ample market headroom is the foundation for capital-market valuation.
Breaking Through the Market: Dual Momentum from Payment Innovation and Global Expansion
At present, the combined market capitalization of China’s top 10 pharmaceutical companies is RMB 1.87 trillion—only 27% of Eli Lilly’s market cap. Compared with leading firms in other domestic industries, Hengrui Medicine, founded in 1970, reported revenue last year that was only about 3% of Huawei and BYD, which were founded roughly 20 years later.
“Innovative drugs account for 81.8% of the market in the United States, 57% in Japan, but only 8.6% in China. In terms of per capita spending on innovative drugs, the U.S. is 124.9 times China, and Japan is 24.4 times China,” said Song Ruilin, Senior President and Chief Expert of the China Association for the Promotion of Medical Innovation. He noted that this enormous market gap directly results in innovative drug prices in China being only about 10% of those in the U.S., making it difficult for companies to generate sufficient profits to sustain growth—one of the key reasons China has struggled to produce a trillion-market-cap pharmaceutical company.
Addressing domestic payment constraints and building a multi-tiered payment system has become a broad industry consensus. Song Ruilin proposed a concrete solution: “Reasonable returns on innovation are the prerequisite for market development. We are not asking for excessive burdens on the basic medical insurance system; rather, we should use commercial health insurance as a breakthrough to build a diversified payment system.”
He suggested that innovative drugs could first be included under medical insurance by referencing the payment standards of comparable indications already covered in the reimbursement catalog, enabling a direct linkage between NMPA approval and the reimbursement list; after two years, they would be re-evaluated based on real-world clinical evidence—if superior, the payment standard would be raised; if equivalent, it would remain unchanged; and if inferior, the drug would be removed from the reimbursement list. “Once cash flow improves for innovative drug companies, it will spur employment, tax revenues, and capital return, forming a virtuous economic cycle of ‘innovation—returns—re-innovation.’ This is the key to high-quality development of China’s pharmaceutical innovation,” he said.
Against the backdrop of tighter medical-insurance spending controls and the normalization of volume-based procurement, commercialization capability has become a core competency for the survival and development of innovative drug companies. Wang Ruwei, Special Assistant to the Chairman of Yangzijiang Pharmaceutical Group, candidly stated during a roundtable discussion: “The current market environment is not optimistic. Homogeneous competition is intense, and in some tracks we even see an ‘involution’ situation where there are no mature products globally, yet more than 100 companies in China are already running clinical trials.”
He added that Yangzijiang Pharmaceutical is seeking breakthroughs through a strategy of “optimizing the existing base and expanding incremental growth”: “We are shifting our marketing model from traditional channel recruitment toward compliant, academically driven promotion, focusing on unmet clinical needs. At the same time, we are accelerating our innovation transformation, with more than 30 innovative drug pipelines in place. In the first half of this year, one Class 1 new drug was approved, and more new products will continue to land in the coming years.”
The localization practices of multinational companies also provide useful references for Chinese firms. Wang Lili, Assistant Vice President of AstraZeneca China, shared: “AstraZeneca entered China in 1993 and built a plant in Taizhou in 2014. Today, Taizhou has become our global supply base for metabolic products. Our products not only serve the China market but are also exported to Europe, Australia, and other regions.” She disclosed that AstraZeneca currently has nine products in the “three highs and three slows” fields, but plans to have at least 18 related products launched by 2034. “We are working with the government and medical institutions to build a one-stop chronic disease management platform, integrating diagnosis and treatment, data, and payment resources, and creating a full-cycle service system from prevention to treatment. This both fits China’s healthcare needs and opens new paths for product commercialization.”
Global expansion has become an inevitable choice for Chinese biopharmaceutical companies to break through domestic market constraints and lift valuations. Wang Junfeng, Co-Chief Investment Officer and Managing Director of Legend Capital, stated: “China’s wave of global partnering and expansion in pharma innovation is showing vigorous development across multiple layers and pathways. In the first half of this year, outbound BD for China’s innovative drugs contributed nearly half of global deal volume.” He emphasized, however, that going global is not simply about product transactions: “Major pharma companies must systematically build global capabilities in the process. Rapidly filling gaps through partnerships, participating in global development, and accumulating experience are the more critical tasks at this stage.”
Xie Xin, Executive Director and Senior Vice President of Sino Biopharmaceutical, clarified the company’s global ambitions: “Our goal is to build a multinational pharma company rooted in China. Over the next five years, we hope to have products approved in the United States and achieve USD 1 billion in annual sales.” He highlighted China’s distinctive advantages: “Our R&D costs are only half of those in the United States, while our R&D speed is double. Coupled with the sense of responsibility and diligence shaped by Confucian culture, Chinese teams are capable of standing out in global competition.”
Beyond mature markets in Europe and the United States, emerging markets such as Southeast Asia have become important breakthrough points for Chinese companies going global. Wang Junfeng noted: “We began strategically deploying in Southeast Asia in 2018, believing it resembles China 15 years ago, with tremendous demographic dividends and market potential.” He explained with an example: “In Indonesia, companies we invested in have already helped products from BeiGene, Junshi Biosciences and other Chinese firms to land locally—whether through bulk drug exports or local commercialization, we have seen positive progress. With 280 million people and strong healthcare demand, Indonesia may become a major overseas market for China’s innovative drugs in the future.”
Capital Restructuring: Ecosystem Evolution from Equity Reliance to BD-Driven Growth
Breakthroughs in technological innovation and the expansion of market布局 depend on efficient capital coordination. China’s biopharmaceutical capital ecosystem is evolving from a single model that relied primarily on equity financing to a diversified ecosystem driven by BD and coordinated across multiple channels, providing sustained momentum for industrial development.
The rise of BD transactions has become a key marker of this shift. “In 2024, the total value of China’s outbound biopharma licensing is expected to exceed USD 100 billion, with upfront payments already surpassing USD 6 billion. More importantly, BD financing has overtaken the primary market and has become the core source of cash flow for many innovative companies,” Wang Junfeng explained further. This shift is reshaping companies’ growth logic: “In the past, companies relied mainly on equity financing. Now, the hard cash generated through BD deals is largely reinvested into R&D, forming a virtuous cycle of ‘R&D—translation—licensing—re-R&D.’ This model not only reduces reliance on equity financing, but also improves the sustainability of R&D.”
Xie Xin, Executive Director of Sino Biopharmaceutical, shared a similar experience: “We have obtained considerable upfront payments through out-licensing. Recently, the acquired company Laixin Pharma just received USD 300 million, and most of these funds were used for R&D. BD income has become an important supplement to our R&D funding, supporting the continuous advancement of our innovative pipeline.”
Hong Kong’s capital market plays an indispensable role in this ecosystem. Yu Changhai, Chairman of the Hong Kong Biotechnology Association, said: “Hong Kong’s Chapter 18A listing regime is an innovation. It has already become Asia’s No. 1 and the world’s No. 2 listing destination for pre-revenue biotech companies. In 2024, Hong Kong’s biopharmaceutical sector rebounded first and became one of the best-performing biopharmaceutical capital markets globally.”
He emphasized that Hong Kong’s value goes beyond financing: “Hong Kong is becoming a cross-border collaborative platform for ‘R&D–financing–translation.’ More and more companies are choosing Hong Kong as a pivot to conduct global fundraising and BD transactions, forming a cross-border innovation ecosystem of ‘Hong Kong financing, mainland R&D, and global translation.’”
Capital’s investment logic is also shifting from “track chasing” to “deep value cultivation.” Cao Yibo, Managing Director of Sequoia China, said: “After the market adjustments of the past few years, capital markets have become more rational in biopharma investing. Differentiation, platformization, and internationalization have become core investment criteria. We adhere to an investment principle of ‘either the first, or the only,’ focusing on scarce innovative assets.”
He further analyzed the core competitiveness of Chinese companies: “China’s R&D costs are only half of those in the United States, while R&D speed is double. Together with indirect cost advantages such as management and facilities, the total cost of Chinese innovative pipelines may be 1/4 to 1/6 of that in the U.S. As overseas companies gradually shed their biases against China’s R&D and clinical data, the global attractiveness of China’s innovative assets will continue to rise.”
Notably, new financing models are injecting fresh vitality into the industry. In a thematic exchange on the digital revolution of RWA and healthcare financing, experts pointed out that RWA tokenization provides a breakthrough pathway for funding pharmaceutical R&D: “By using blockchain technology to convert real-world assets such as future revenue rights of R&D pipelines and medical data into tradable digital tokens, the logic shifts from ‘selling equity’ to ‘selling expectations.’” This model can effectively unlock idle assets, enabling global capital to participate directly in early-stage innovation and share returns. Experts also stressed that, to ensure implementation, compliant infrastructure such as a “regulatory sandbox–stablecoin–exchange” framework must be established, while addressing critical challenges including on-chain data authenticity, cross-border regulatory coordination, and system security.
Policy support is further improving the capital ecosystem. Song Ruilin called for: “We call for a Huawei or BYD in pharmaceuticals, but that requires policy support and market inclusiveness. We need to improve the patent term extension/compensation system to prevent the patent cliff from arriving prematurely. China has already established a patent term compensation mechanism through reforms of the drug review and approval system, which, in theory, can provide up to 14 years of post-launch patent protection. However, due to the pressure of a single payment system, prices of many innovative drugs fall rapidly after launch, bringing the patent cliff forward by at least 10 years. This severely undermines companies’ incentives to innovate and weakens investors’ confidence.”
He also suggested: “A dedicated registration classification for natural products should be established. China has unique advantages in natural products, but the current classification system makes it difficult for such innovation to obtain reasonable pricing and market recognition, constraining industry development.”
As China advances from a “major pharmaceutical country” to a “pharmaceutical powerhouse,” Wang Junfeng expressed confidence in the future: “After ten years of development, China’s innovative drugs have moved from quantitative change to qualitative change. Driven by efficiency, talent, infrastructure, and market potential, China will play a more important role globally over the next decade. We look forward to accompanying more Chinese innovative drug companies to secure a place in the global arena of innovative medicines and to write a value story that belongs to China’s biopharmaceutical industry.”
The global community is awaiting China’s next-generation innovative medicines.
This July, China’s pharmaceutical community was ignited by a blockbuster deal worth tens of billions of dollars.
Hengrui Pharma struck a USD 12.5 billion partnership with global pharmaceutical giant GlaxoSmithKline to co-develop up to 12 innovative medicines, setting a new record for China’s innovative-drug “going global” efforts.
Just one month earlier, China’s homegrown innovative drug Zelemi® (benvitimod cream) had also set a record.
The American Academy of Dermatology (AAD)—widely regarded as the global bellwether in dermatology—broke its all-time fastest revision record for the gold-standard treatment of atopic dermatitis, updating the guideline within 23 months and listing Zelemi® (benvitimod cream) in the highest tier as “strongly recommended.”
This means that, for the first time, dermatologists across 160 countries and regions are explicitly guided by the guideline to prioritize a Chinese medicine when treating eczema.
From the recent surge in transactions to Zelemi®’s pioneering exploration, Chinese pharmaceutical companies are providing new medicines for the world.
Breaking Records
A record has been broken!
In the first half of 2025, the total value of China’s innovative-drug license-out transactions reached nearly USD 66 billion—surpassing the full-year total of last year and setting a new record for China’s innovative drugs going global.
Innovative-drug BD deals have been booming, with deal values repeatedly hitting new highs.
On May 20, the upfront payment in Sansheng’s partnership with Pfizer reached USD 1.25 billion, with a total deal value exceeding USD 6 billion—setting a new record for the upfront payment of a domestically developed innovative drug.
On July 28, Hengrui Pharma signed a strategic cooperation agreement with GlaxoSmithKline to co-develop up to 12 innovative medicines, with an upfront payment of USD 500 million and a total potential value of up to USD 12 billion.

On October 22, Innovent Biologics reached a collaboration with Takeda on next-generation IO and ADC therapies, with an upfront payment of USD 1.2 billion and a total deal value of up to USD 11.4 billion.
BD stands for Business Development, referring to commercial activities through which pharmaceutical companies integrate resources, replenish pipelines, expand markets, or share risks via collaborations, licensing, M&A, technology transfers, and related approaches.
Licensing/out-licensing of innovative drugs is one of the most common forms of BD.
In the past, Chinese pharmaceutical companies mainly “brought in” medicines—purchasing products from multinational drugmakers.
Today, Chinese pharmaceutical companies are “going out” and expanding into international markets.
China’s innovative drugs are delivering more and better treatment options to the world.
Amid this wave, one Chinese innovative drug targeting dermatologic diseases has rewritten the treatment dilemma for hundreds of millions of patients.
Atopic dermatitis, commonly known as “eczema,” affects nearly 400 million people worldwide and is the largest disease category in dermatology.
Notably, about 10%–20% of children worldwide suffer from eczema.
Data show that the prevalence among infants under 1 year old in China is 30%, and the prevalence among children aged 1 to 7 is 13%.
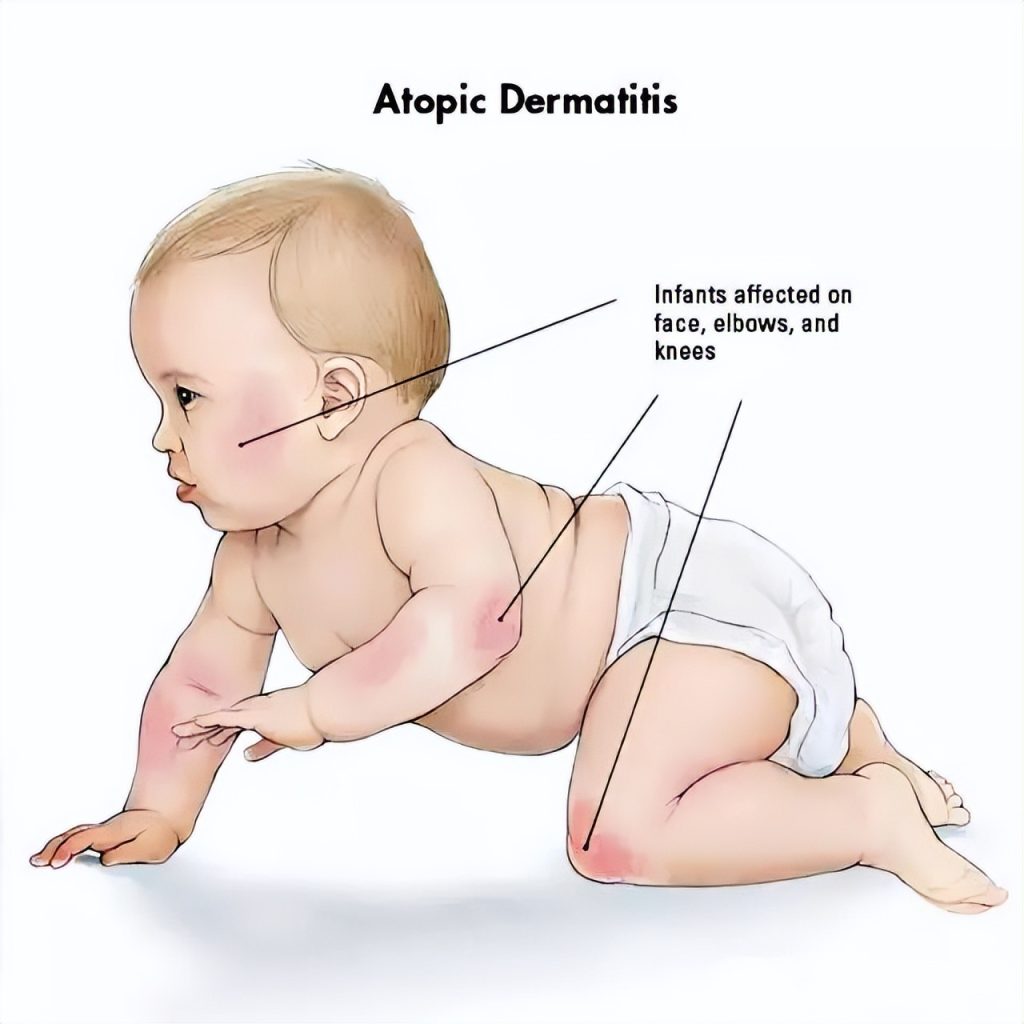
Although eczema is not fatal, it torments patients with recurrent, intense itching. In severe cases, it can even affect growth and development in children under 12, leading to insomnia, changes in appearance, and other issues—substantially reducing quality of life.
For a long time, eczema treatment has been trapped in a “double bind.”
More than 90% of patients use topical therapies. One option is corticosteroids, which act quickly but carry risks such as skin atrophy and hyperpigmentation with long-term use. Another option is topical calcineurin inhibitors, which have a favorable safety profile but tend to have a slower onset and weaker efficacy.
Children have a fragile skin barrier and are prone to relapse. Coupled with caregivers’ concerns about side effects, adherence is often poor—creating a cycle in which “without medication the child suffers, but with medication there are lingering worries.”
The range of topical options for eczema is very limited and does not adequately meet pediatric treatment needs.
It was not until 2024 that patients worldwide finally saw a new medicine.
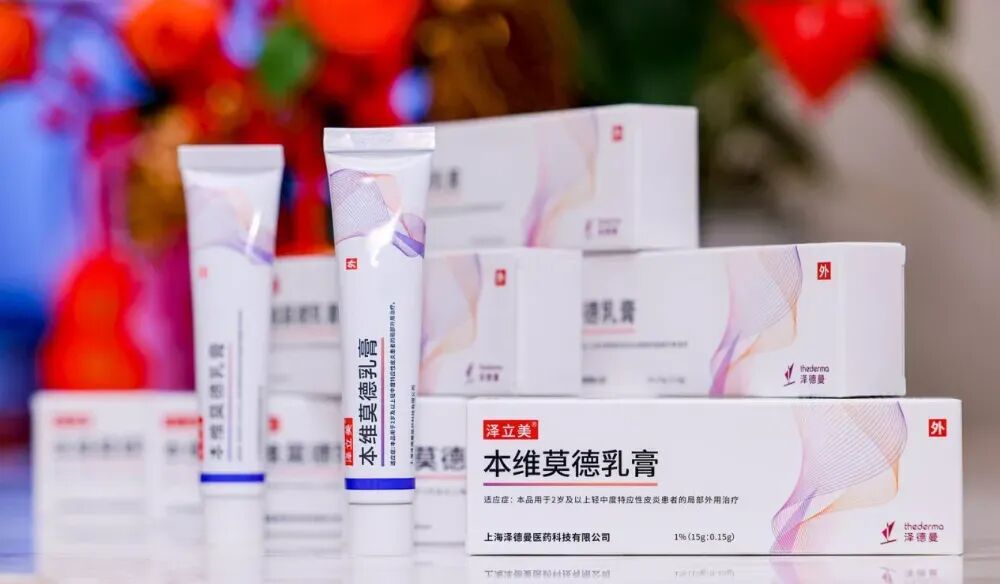
On November 22, 2024, Zelemi® (benvitimod cream), independently developed by Shanghai Zedeman Pharmaceutical Technology Co., Ltd., was approved for marketing by China’s National Medical Products Administration (NMPA).

Just 20 days later, Zelemi® (benvitimod cream) was also approved by the U.S. FDA, achieving a simultaneous first launch in China and the United States.
Zelemi® (benvitimod cream) is a homegrown innovative drug with a brand-new mechanism of action and target. It is also the world’s first aryl hydrocarbon receptor (AhR) modulator approved for the treatment of eczema (atopic dermatitis) in children aged 2 years and older as well as adults.
As a non-steroidal topical therapy, clinical results for Zelemi® (benvitimod cream) show that itching can stop as early as the day after treatment begins. One year after discontinuation, 70% of patients did not relapse. Systemic exposure risk is extremely low, and the main adverse reaction is only mild folliculitis.
Achieving rapid onset, strong efficacy, and durable benefit simultaneously among non-steroidal therapies, this outstanding therapeutic performance drew the attention of the American Academy of Dermatology (AAD).
The AAD’s guideline updates are known for rigor: after the 2014 edition was released, it took nine years to complete the revision in 2023.
Tapinarof is the overseas name for benvitimod.
After confirming the clinical value of Zelemi® (benvitimod cream), the AAD completed an update in the shortest cycle on record and issued a “strong recommendation.”
To date, Zelemi® (benvitimod cream) has been approved for marketing in multiple countries, including China, the United States, Japan, Canada, Germany, and France.
China’s innovative drugs are bringing new therapies and new choices to hundreds of millions of patients worldwide.
A 30-Year Marathon
Innovative drug R&D has long been described as a “high-risk adventure with slim odds”—difficult, risky, time-consuming, and highly specialized.
From early research to final launch, an innovative drug typically takes 10–15 years on average, requires an investment of USD 2–3 billion, and has a clinical-stage success rate of less than 10%.
The birth of Zelemi® (benvitimod cream) is precisely the story of nearly 30 years of perseverance and breakthroughs in this “adventure.”
In the 1990s, Chen Genghui, who was pursuing a PhD in biology at Simon Fraser University in Canada, isolated—together with his team members—a stilbene-like small-molecule compound from metabolites of a symbiotic bacterium associated with a soil nematode.

This compound later became benvitimod (English name: Tapinarof), which would go on to make waves across the global dermatology field.
Although benvitimod has a molecular weight of only 254 Da, it can precisely modulate the aryl hydrocarbon receptor (AhR).
The aryl hydrocarbon receptor (AhR) is an intracellular, ligand-activated transcription factor widely expressed in skin keratinocytes, immune cells, and epithelial cells.
If the human immune system is a house, the AhR is the master sensor that can control all the lights.
Once activated, it triggers a cascade of immune responses.
Chen Genghui and his team members keenly realized that activating and modulating this “super target” via benvitimod could regulate cells involved in skin inflammation, thereby suppressing inflammation and oxidative stress and promoting restoration of skin barrier function.
Even more importantly, benvitimod also exhibits good skin permeability.
Based on its biological properties, benvitimod is well suited to be formulated as a topical medicine for the treatment of skin diseases.
In 1999, Chen Genghui, together with his classmate Li Jianxiong and their mentor Webster, formally founded a company in Canada, obtained the patent grant for benvitimod, and embarked on a systematic R&D journey.
Later, Chen Genghui returned to China to continue advancing benvitimod’s preclinical research and subsequent clinical trials.
In May 2019, benvitimod cream was approved for marketing for the treatment of mild-to-moderate plaque psoriasis in adults.
In 2021, Chen Genghui founded Zedeman Pharma in Shanghai’s Pudong New Area, embarking on his “third entrepreneurship” and launching the “second development” of benvitimod.

This time, Chen Genghui aimed to develop an AhR modulator based on benvitimod cream to tackle atopic dermatitis (eczema), the largest disease category in dermatology.
In November 2024, Zelemi® (benvitimod cream) achieved “dual submission and dual approval” in China and the United States, marking a milestone in China’s new-drug globalization.
The R&D characteristics of “high investment, high risk, high failure rate, and long cycle” determine that innovative drugs generally carry relatively high prices in their early stages.
However, Zelemi® (benvitimod cream) adhered to an affordable, patient-friendly pricing strategy, with its initial domestic price set at only one-tenth of the U.S. price.
To further improve access, Zedeman Pharma continued to optimize manufacturing efficiency, control costs, and upgrade its management system, cutting the price by 60% in July 2025.
At the same time, Zedeman Pharma also proactively participated in medical-insurance negotiations and passed the initial review for inclusion in the national reimbursement program.
Looking ahead, as policy dividends such as reimbursement inclusion are realized, people in China will be able to access top-tier innovative medicines with internationally comparable quality and efficacy at more affordable prices.
One molecule, two world-class breakthroughs!
Chen Genghui and his team have upheld innovation for 30 years—achieving success across two indications for a single drug and establishing a benchmark for China’s innovative drugs, from first launch to global recognition.
A Methodology for Innovation
From a compound in the laboratory to an innovative drug in the global spotlight, Zelemi’s innovation journey offers a replicable “methodology of innovation” for China’s new-drug R&D.
A needs-driven approach is innovation’s “compass.”
At the core of innovation is responding to real clinical needs.
Zelemi’s success stems from a precise understanding of the therapeutic dilemma in pediatric atopic dermatitis.
“Innovation can’t be innovation for its own sake—you have to first clarify where the real need lies.”
Chen Genghui admitted that his own child once suffered from eczema, and he personally witnessed the urgent clinical demand for medications suitable for children with atopic dermatitis.
This empathy and insight—rooted in lived experience—strengthened Chen Genghui’s determination to pursue the R&D direction.
This offers a lesson for Chinese pharmaceutical companies: innovation should be grounded in frontline clinical practice, anchored in patient pain points, and driven first and foremost by real needs.
Patent strategy is innovation’s “protective shield.”
For the new indication of atopic dermatitis, Zelemi built a full-chain patent matrix—from core compound patents to formulation processes, manufacturing workflows, and indication expansion—forming a tightly woven IP protection network.
This not only ensures that innovative are protected, enabling sustained investment in R&D, but also ultimately benefits more patients worldwide.
In the innovative-drug arena, the depth and breadth of patent directly determine a company’s core competitiveness.
Long-termism is innovation’s “star chart.”
From isolating the compound in the 1990s to Zelemi’s launch in 2024, Chen Genghui and his team’s innovation journey spanned nearly 30 years—through funding shortages and team turbulence—yet they consistently core technology R&D.
This resolve to “forge a sword over ten years” is precisely the quality China’s new-drug R&D needs most today.
There are no “shortcuts” in innovative-drug R&D—only steadfast commitment and long-term investment can nurture heavyweight that withstand clinical and market validation.
An ecosystem is innovation’s “accelerator.”
Although benvitimod was discovered in Canada, China achieved its complete translation from the laboratory to clinical application.
Zelemi’s success is inseparable from comprehensive support across China’s biopharmaceutical industry ecosystem.
One telling detail: Zelemi’s marketing application was accepted in China in December 2023, included in priority review in January 2024, and approved for marketing in November of the same year.
From clinical development to approval, China took only 523 days, while the United States took 1,198 days—China was more than twice as fast.
Behind this are the policy dividends unleashed by China’s drug review and approval reforms, as well as coordinated efforts enabled by a mature industry ecosystem.
Take Shanghai’s Pudong New Area—home to Zedeman Pharma—as an example: its biopharmaceutical industry has reached RMB 410 billion,more than 4,000 biopharma enterprises, with 718 innovative-drug pipelines under development—about one-fifth of the national total and 6% globally. The cumulative number of approved domestically developed innovative drugs and Class III innovative medical devices accounts for 13% and 11% of the national total, respectively.

From basic research and incubation/translation, to clinical trials, review and approval, and then to manufacturing deployment and, the “full-chain innovation ecosystem” built in Pudong New Area provided comprehensive support for Zelemi’s R&D and launch—spanning talent, capital, technology, and policy.
Zelemi’s success is the result of Zedeman Pharma’s on a needs-driven approach and deep cultivation of core technologies; it is also an inevitability stemming from China’s increasingly biopharma ecosystem and the coordinated force of policy and the market.
This provides a highly valuable practical model for China’s new-drug R&D and signals that China’s innovative drugs are moving from “single-point breakthroughs” toward a new stage of “systematic rise.”
A Steady Stream of Breakthroughs
Zelemi’s innovation legend is a vivid microcosm of China’s rise in new-drug development.
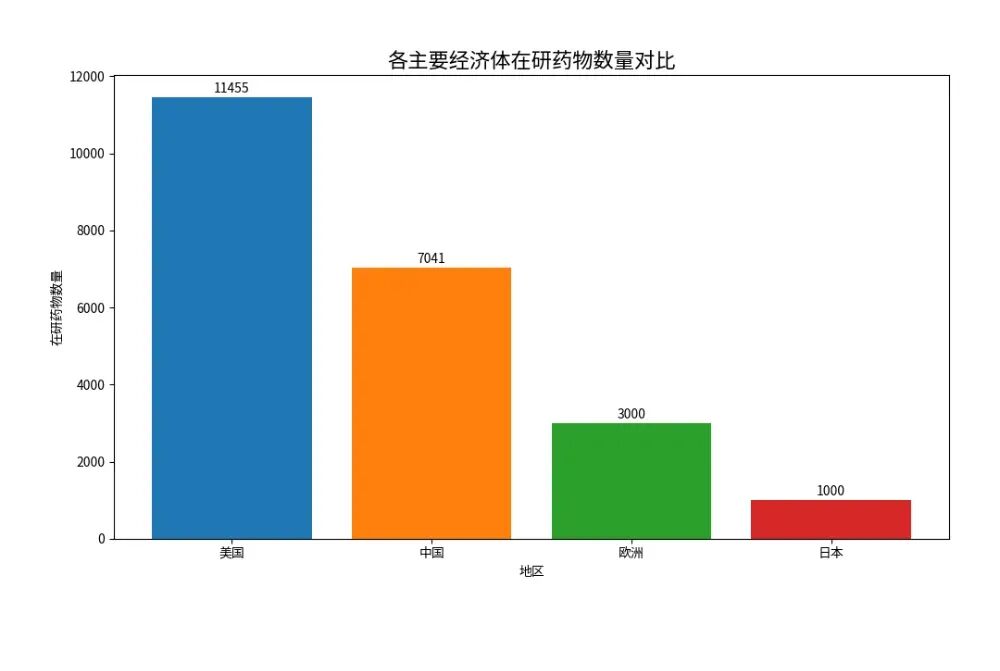
From 2015 to 2025, the number of drugs under development in China surged from 851 to 7,041 over a decade, representing a compound annual growth rate of 23.5%—3.4 times the global average (6.9%).
Even more noteworthy, China’s first-in-class drugs surged from 9 in 2015 to 120 in 2024, with its global share rising from 9% to 31.3%—ranking second worldwide and rapidly closing in on the United States’ leading position.
As Dr. Chen Genghui, inventor of benvitimod and Chairman of Shanghai Zedeman Pharmaceutical Technology Co., Ltd., put it: In the past, Chinese people waited for Western new drugs; today, the world is waiting for Chinese new drugs.
This assessment has been fully validated by the market: in the first half of 2025, the total value of China’s innovative-drug license-out transactions reached nearly USD 66 billion, surpassing last year’s full-year total.
Multinational pharmaceutical giants such as Pfizer, GlaxoSmithKline, and Takeda are spending heavily to secure China’s innovative assets, voting with real money on China’s original R&D strength.
The profound transition from “follow-on innovation” to “source innovation” is propelling China’s pharmaceutical innovation into a golden era of rising quality and efficiency—and will reshape the global balance of “voice” in healthcare.
Looking ahead, more innovative drugs like Zelemi—addressing unmet clinical needs and setting global treatment directions—will continue to emerge.
The Chinese new drugs the world is waiting for are accelerating toward arrival.
M.U.S.T. Academician Zhu Yizhun Wins First Place in the Biomedical Start-up Group at the 13th China Innovation & Entrepreneurship Competition
Held from November 18 to 21, 2024, in Taizhou, Jiangsu, the National Finals of the 13th China Innovation & Entrepreneurship Competition operated under the theme “Gathering for Innovation, Moving Towards the New.” The project titled “Discovery and Clinical Research of Original Anti-inflammatory and Immune Drugs,” led by Academician Zhu Yizhun—Associate Vice-President of M.U.S.T. and Dean of the School of Pharmacy—secured First Place in the Biomedical Start-up Category. This victory, achieved through substantial research results and outstanding innovation, represents the competition’s highest honor.

Since its launch in June, the competition has attracted over 10,000 teams led by professors and overseas-returned PhDs. Following intense rounds across 37 regional divisions, 204 teams advanced to the national biomedical finals (56 in the Start-up Group and 148 in the Growth Group). Entries spanned cutting-edge fields such as innovative drugs, synthetic biology, medical devices, AI healthcare systems, stem cell therapies, and mRNA vaccines. Ultimately, only six start-up teams and 14 growth-stage teams emerged from the semi-finals to compete in the championship round.
During the finals, teams showcased innovative concepts and technologies in areas such as immune drug development, cancer screening, and toxicology. A panel of judges conducted comprehensive evaluations based on technological innovation, business models, market prospects, team capability, and financial health. Distinguished by its solid research foundation and exceptional innovative potential, Academician Zhu’s project stood out among the competitors to claim the top prize.

Supported by the Science and Technology Development Fund (FDCT) of Macau, the project leverages the technological platforms of the State Key Laboratory of Quality Research in Chinese Medicine (M.U.S.T.) and the Laboratory for Natural Drug Discovery and Industrialization. This accolade provides significant momentum for the team’s future development. Moving forward, the team plans to deepen its research and collaborate with various institutions to accelerate the translation of these innovative findings into clinical applications.







