Product R&D
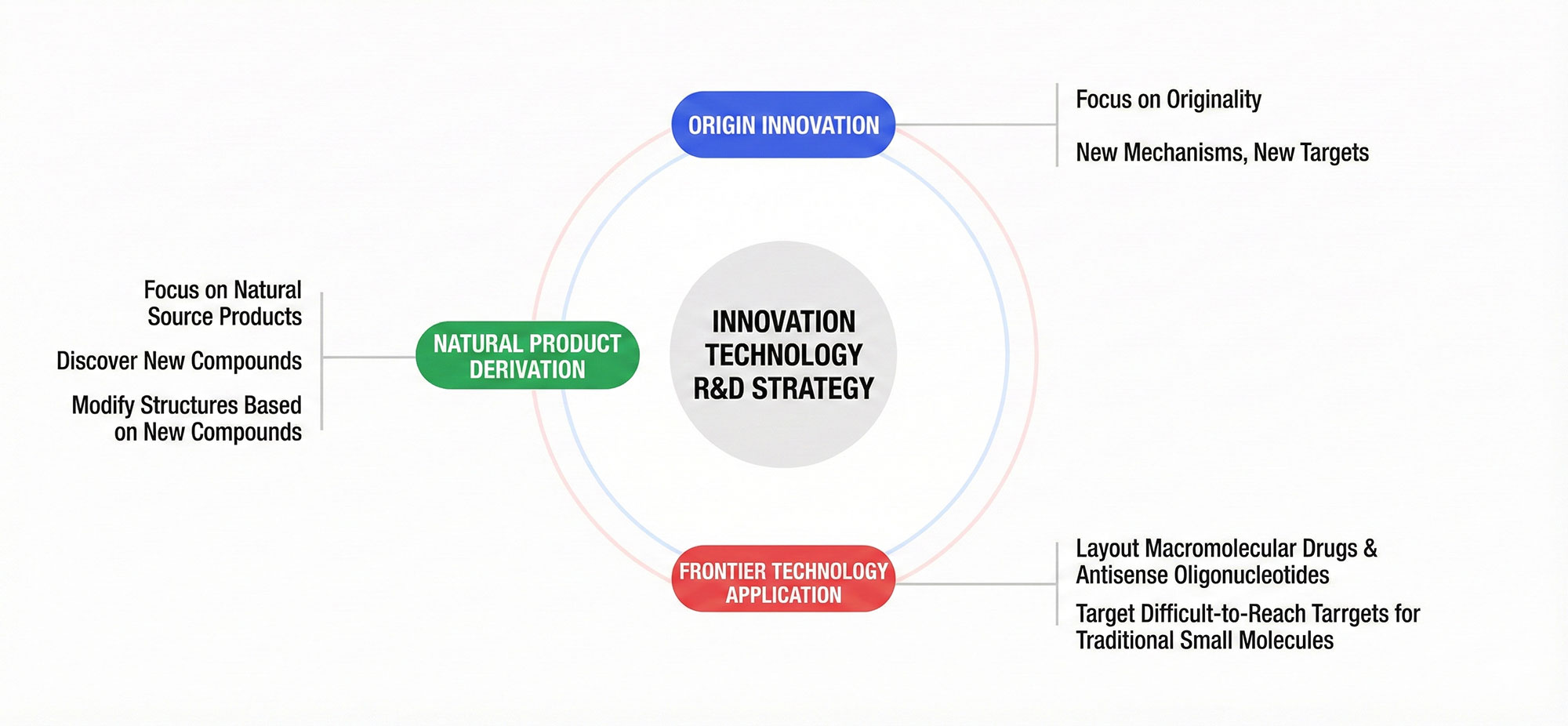
R&D Strategy
Leveraging strong scientific capabilities and support from world-class academic resources, InnoDrug implements a multi-pronged R&D strategy across multiple dimensions. Our research primarily focuses on novel compounds from natural sources and their subsequent optimization and structural refinement. We continuously explore ways to enhance the performance of new drugs across different indications, and aim to earn patient and market recognition through innovative mechanisms of action and novel therapeutic targets.
Product Pipeline
InnoDrug advances three major product pipelines. Our 20 in-house discovered innovative drug candidates span Phase I/II clinical studies, IND-enabling, and preclinical research, ensuring a robust and sustainable portfolio to support future launches.
Cardiovascular & Cerebrovascular
Oncology
Metabolism & Inflammatory Immunology
| No. | Therapeutic Area | Program / Candidate | Target | Indication | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Cardiovascular & Cerebrovascular | Leonurine Sulfate Tablets (SCM-198) | Nox4 | Hyperlipidemia | Phase I | Phase II | Phase III | Launch | |||
| 2 | Cardiovascular & Cerebrovascular | ZYZ-451 | SOD/CAT | Heart Failure | Preclinical | IND | Phase I | Phase II | Phase III | ||
| 3 | Cardiovascular & Cerebrovascular | INDL-001 (HDAC4) | HDAC4 | Vascular Inflammation | Preclinical | IND | Phase I | Phase II | Phase III | ||
| 4 | Cardiovascular & Cerebrovascular | INDL-002 (JMJD3) | JMJD3 | Vascular Injury | Preclinical | IND | Phase I | Phase II | Phase III | ||
| 5 | Cardiovascular & Cerebrovascular | INDL-006 AS (Fra1) | Fra1 |
Vascular Aging
(Upstream Large Molecule) |
Preclinical | IND | Phase I | Phase II | |||
| 6 | Cardiovascular & Cerebrovascular | INDL-005 (GATA-4) | GATA-4 |
Vascular Aging
(Midstream Large Molecule) |
Preclinical | IND | Phase I | Phase II | |||
| 7 | Cardiovascular & Cerebrovascular | INDL-005 (GATA-4) | GATA-4 |
Vascular Aging
(Downstream Large Molecule) |
Preclinical | IND | Phase I | Phase II | |||
| 8 | Cardiovascular & Cerebrovascular | INDL-010 (HDAC4/SMYD3 Double AS) |
HDAC4/
SMYD3 |
Vascular Aging
(Double AS) |
Preclinical | IND | Phase I | ||||
| 9 | Oncology | ZYZ-384 | SMYD3 | Hepatocellular Carcinoma | Preclinical | IND | Phase I | Phase II | Phase III | ||
| 10 | Oncology | ZYZ-410 | HO-1 | Non-Small Cell Lung Cancer | Preclinical | IND | Phase I | Phase II | |||
| 11 | Oncology | YL-B11 | DDR1 |
Pancreatic Cancer
(DDR1) |
Preclinical | IND | Phase I | Phase II | |||
| 12 | Oncology | YL-B1102 |
Blocks DDR1, CTLA-4, and PD-L1; Activates CD28 |
ADC and Multi-Target Large-Molecule Antitumor Therapy (CTLA-4, PD-L1) |
Preclinical | IND | Phase I | Phase II | |||
| 13 | Metabolism & Inflammatory Immunology | S-Propargyl-Cysteine Capsules (ZYZ-802) | CSE | Rheumatoid Arthritis | IND | Phase I | Phase II | Phase III | Launch | ||
| 14 | Metabolism & Inflammatory Immunology | Uncaria Alkaloid (Gou-Teng Alkaloid) |
PI3K/Akt/
GSK3β Pathway |
Insulin Resistance, Diabetes, and Related Complications |
Preclinical | IND | Phase I | Phase II | |||
| 15 | Metabolism & Inflammatory Immunology | M2-lipo SCM-198 | MMP-9 |
Ulcerative Colitis
(Leonurine Alkaloid Nanoformulation) |
Preclinical | IND | Phase I | Phase II | |||
| 16 | Metabolism & Inflammatory Immunology | ZYZ-488 | MMP-9 | Dysmenorrhea and Premature Ovarian Insufficiency | Preclinical | IND | Phase I | Phase II | |||
| 17 | Metabolism & Inflammatory Immunology | Leonurine Sulfate |
Specific molecules (TNF-α, IL-6, MMP-2, MMP-9) and signaling pathways (VEGF pathway) |
Endometriosis | Preclinical | IND | Phase I | Phase II | |||
| 18 | Metabolism & Inflammatory Immunology | Allyl Cysteine Capsules | Nrf2/NF-κB | Infectious Diarrhea | Preclinical | IND | Phase I | Phase II | |||
| 19 | Metabolism & Inflammatory Immunology | Leonurine Sulfate Gel |
Simultaneously inhibits pro-inflammatory pathways (JAK-STAT, PI3K-Akt) and activates pro-repair pathways (mTOR, ERK) |
Wound Healing | Preclinical | IND | Phase I | Phase II | Phase III | ||
| 20 | Metabolism & Inflammatory Immunology | S-Propargyl-Cysteine Gel | – |
Diabetic Foot Wound Repair and Skin Regeneration |
Preclinical | IND | Phase I | Phase II | Phase III | ||
Product Patents
| No. | Product | Patent Status |
|---|---|---|
| 1 | Leonurine Sulfate (SCM-198) | ✅ Granted patents: 13 in China, 1 in the EU (plus 1 pending), 1 in Japan, and 1 PCT patent |
| 2 | S-Propargyl-Cysteine (ZYZ-802) | ✅ Granted patents: 11 in China, 1 in the U.S., and 1 PCT patent; 2 applications pending |
| 3 | ZYZ-384 | ✅ 1 granted patent in China |
| 4 | ZYZ-451 | ✅ 2 granted patents in China |
| 5 | Uncaria Alkaloid (Gou-Teng Alkaloid) | ✅ 1 granted patent in China |
| 6 | YL-B11 | ✅ 1 granted patent in China |
| 7 | YL-B1102 | ✅ 1 granted patent in China |
| 8 | HDAC4 Inhibitor (INDL-001) | ✅ 2 patent applications pending |
| 9 | JMJD3 Inhibitor (INDL-002) | ✅ 1 application filed in China; 2 applications pending |
| 10 | SMYD3 Inhibitor (INDL-003) | ✅ 1 application filed in China; 2 applications pending |
| 11 | FRA-1 Inhibitor (INDL-004) | ✅ 2 patent applications pending |
| 12 | GATA-4 Inhibitor (INDL-005) | ✅ 2 patent applications in preparation |
Patent Certificate Gallery




Indication Coverage
Our 20 programs under development address 33 indications, offering substantial commercialization potential.

Leonurine Sulfate
- Hyperlipidemia
- Endometriosis
- Ulcerative Colitis
- Wound Healing
- Nonalcoholic Fatty Liver Disease (NAFLD)
- Atherosclerosis

S-Propargyl-Cysteine
- Rheumatoid Arthritis
- Infectious Diarrhea
- Diabetic Foot Wound Repair and Skin Regeneration
- Alzheimer’s Disease
- Myocardial Injury

Small molecule Drugs
- Liver Cancer
- Non-Small Cell Lung Cancer
- Heart Failure
- Anti-Myocardial Ischemia
- Vascular Endothelial Aging
- Insulin Resistance, Diabetes, and Related Complications
- Dysmenorrhea and Premature Ovarian Insufficiency
- Colitis
- Neuroinflammatory Disorders
- Atopic Dermatitis
- Cognitive Impairment

Macromolecular Drugs
- Solid Tumors
- Triple-Negative Breast Cancer (TNBC)
- Gastric Cancer
- NSCLC
- Pulmonary Fibrosis
- Liver Fibrosis
- Atherosclerosis
- Vascular Inflammation
- Vascular Injury
- Vascular Aging



